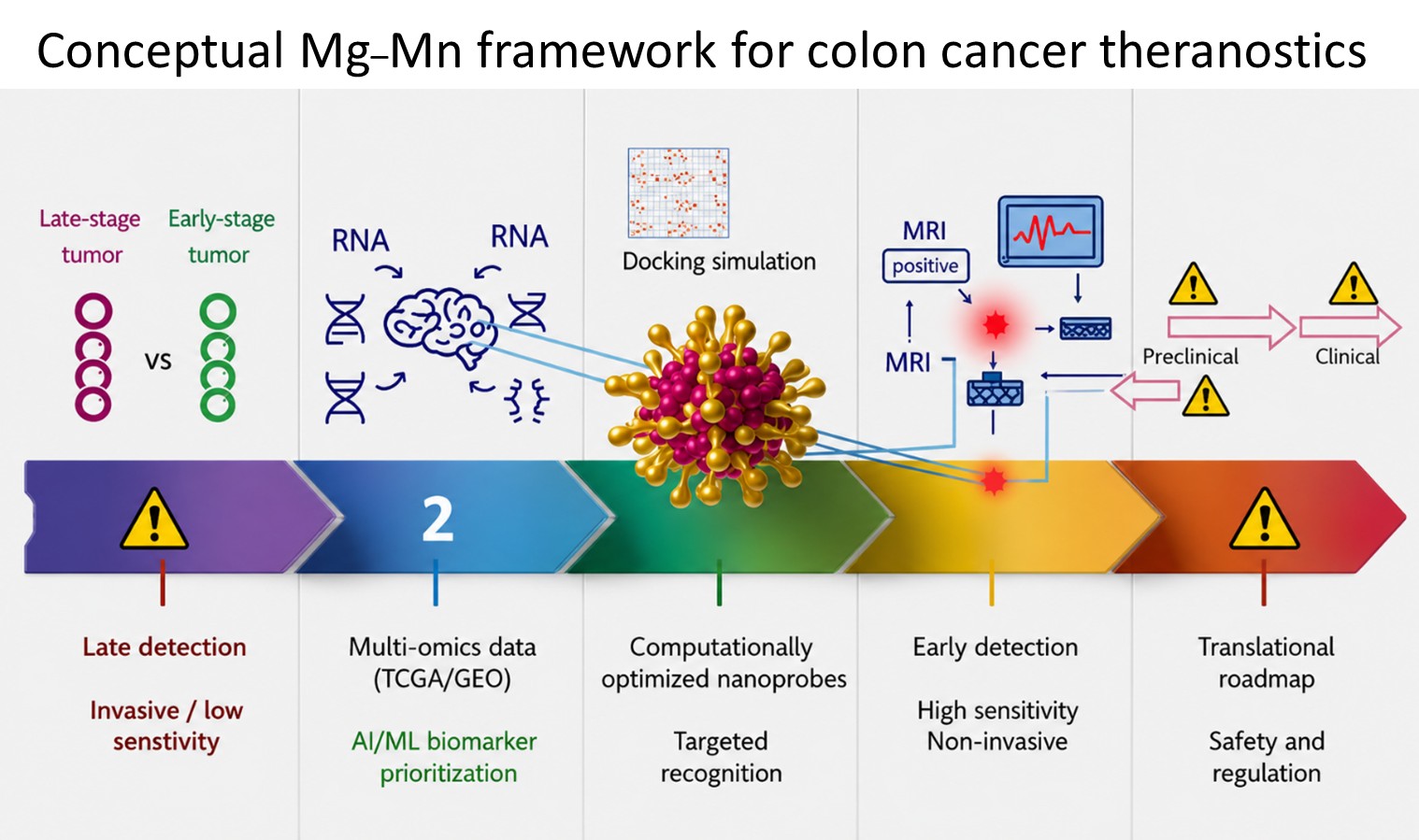
Computational and nanotechnology strategies for colorectal cancer detection and therapy: A narrative review and conceptual framework for magnesium–manganese nanoparticles

Colorectal cancer (CRC) remains a leading cause of global cancer-related mortality, where patient prognosis is heavily dictated by the timing of detection. While CRC encompasses both colon and rectal malignancies, this narrative review focuses primarily on the early detection of colon tumors, which represent the majority of CRC cases and serve as a primary target for biomarker-driven strategies. Despite the utility of conventional screening methods such as colonoscopy, limitations regarding invasiveness and accessibility persist. This review synthesizes interdisciplinary progress from 2019 to 2025, highlighting how the convergence of computational biology and nanotechnology offers a promising path forward. Computational strategies, particularly machine learning and omics analysis, have accelerated the discovery of predictive biomarkers. In parallel, nanotechnology platforms—including gold nanoparticles and quantum dots—have emerged as high-sensitivity tools for biosensing. A central feature of this review is a conceptual framework on magnesium–manganese nanoparticles, which provides a theoretical framework for their dual roles as biosensors and imaging agents. This framework underscores the translational potential of combining in silico biomarker discovery with nanomaterial design. Finally, the review examines the hurdles to clinical translation and outlines a roadmap for using artificial intelligence-driven optimization to achieve precise, non-invasive early detection of colon-specific malignancies across the CRC spectrum.
- Rawla P, Sunkara T, Barsouk A. Epidemiology of colorectal cancer: incidence, mortality, survival, and risk factors. Prz Gastroenterol. 2019;14(2):89-103. doi: 10.5114/pg.2018.81072
- Cardoso R, Guo F, Heisser T, et al. Colorectal cancer incidence, mortality, and stage distribution in European countries in the colorectal cancer screening era: an international population-based study. Lancet Oncol. 2021;22(7):1002-1013. doi: 10.1016/S1470-2045(21)00199-6
- Morgan E, Arnold M, Gini A, et al. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut. 2023;72(2):338-344. doi: 10.1136/gutjnl-2022-327736
- Lakemeyer L, Sander S, Wittau M, Henne-Bruns D, Kornmann M, Lemke J. Diagnostic and prognostic value of CEA and CA19-9 in colorectal cancer. Diseases. 2021;9(1):21. doi: 10.3390/diseases9010021
- Rao H, Wu H, Huang Q, Yu Z, Zhong Z. Clinical value of serum CEA, CA24-2 and CA19-9 in patients with colorectal cancer. Clin Lab. 2021;67(4):1079-1089. doi: 10.7754/Clin.Lab.2020.200828
- Sefrioui D, Beaussire L, Gillibert A, et al. CEA, CA19-9, circulating DNA and circulating tumour cell kinetics in patients treated for metastatic colorectal cancer (mCRC). Br J Cancer. 2021;125(5):725-733. doi: 10.1038/s41416-021-01431-9
- Luo H, Shen K, Li B, Li R, Wang Z, Xie Z. Clinical significance and diagnostic value of serum NSE, CEA, CA19-9, CA125 and CA242 levels in colorectal cancer. Oncol Lett. 2020;20(1):742-750. doi: 10.3892/ol.2020.11633
- Ahmed M. Colon cancer: a clinician’s perspective in 2019. Gastroenterol Res. 2020;13(1):1-10. doi: 10.14740/gr1239
- Combes GF, Vučković AM, Perić Bakulić M, et al. Nanotechnology in tumor biomarker detection: the potential of liganded nanoclusters as nonlinear optical contrast agents for molecular diagnostics of cancer. Cancers. 2021;13(16):4206. doi: 10.3390/cancers13164206
- Nasrollahpour H, Khalilzadeh B, Hasanzadeh M, et al. Nanotechnology-based electrochemical biosensors for monitoring breast cancer biomarkers. Med Res Rev. 2023;43(3):464-569. doi: 10.1002/med.21931
- Mann M, Kumar C, Zeng WF, Strauss MT. Artificial intelligence for proteomics and biomarker discovery. Cell Syst. 2021;12(8):759-770. doi: 10.1016/j.cels.2021.06.006
- Pan D, Schmieder AH, Wickline SA, Lanza GM. Manganese-based MRI contrast agents: past, present and future. Tetrahedron. 2011;67(44):8431-8444. doi: 10.1016/j.tet.2011.07.076
- Agarwal S, Curtin J, Duffy B, Jaiswal S. Biodegradable magnesium alloys for orthopaedic applications: a review on corrosion, biocompatibility and surface modifications. Mater Sci Eng C Mater Biol Appl. 2016;68:948-963. doi: 10.1016/j.msec.2016.06.020
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71
- Baethge C, Goldbeck-Wood S, Mertens S. SANRA—a scale for the quality assessment of narrative review articles. Res Integr Peer Rev. 2019;4:5. doi: 10.1186/s41073-019-0064-8
- Socovich AM, Naba A. The cancer matrisome: from comprehensive characterization to biomarker discovery. Semin Cell Dev Biol. 2019;89:157-166. doi: 10.1016/j.semcdb.2018.06.005
- Leclercq M, Vittrant B, Martin-Magniette ML, et al. Large-scale automatic feature selection for biomarker discovery in high-dimensional OMICs data. Front Genet. 2019;10:452. doi: 10.3389/fgene.2019.00452
- Qi X, Lin Y, Chen J, Shen B. Decoding competing endogenous RNA networks for cancer biomarker discovery. Brief Bioinform. 2020;21(2):441-457. doi: 10.1093/bib/bbz081
- Ng S, Masarone S, Watson D, Barnes MR. The benefits and pitfalls of machine learning (ML) for biomarker discovery. Cell Tissue Res. 2023;394(1):17-31. doi: 10.1007/s00441-023-03816-z
- Tomczak K, Czerwińska P, Wiznerowicz M. The Cancer Genome Atlas (TCGA): an immeasurable source of knowledge. Contemp Oncol. 2015;19(1):68-77. doi: 10.5114/wo.2014.47136
- National Cancer Institute. Citing TCGA in publications and presentations. The Cancer Genome Atlas. Updated March 6, 2019. Available from: https://www.cancer.gov/ccg/research/ genome-sequencing/tcga/using-tcga-data/citing [Last accessed on September 11, 2025].
- National Center for Biotechnology Information. Gene Expression Omnibus (GEO). Published 2019. Available from: https://www.ncbi.nlm.nih.gov/geo/ [Last accessed on September 11, 2025].
- European Bioinformatics Institute. ArrayExpress: functional genomics data. EMBL-EBI. Published March 6, 2019. Available from: https://www.ebi.ac.uk/biostudies/ arrayexpress [Last accessed on September 11, 2025].
- Robinson MD, McCarthy DJ, Smyth GK. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 2010;26(1):139- 140. doi: 10.1093/bioinformatics/btp616
- Anders S, Huber W. Differential expression analysis for sequence count data. Genome Biol. 2010;11(10):R106. doi: 10.1186/gb-2010-11-10-r106
- Liu S, Wang Z, Zhu R, Wang F, Cheng Y, Liu Y. Three differential expression analysis methods for RNA sequencing: limma, EdgeR, DESeq2. J Vis Exp. 2021;(175):e62528. doi: 10.3791/62528
- Reimand J, Isserlin R, Voisin V, et al. Pathway enrichment analysis and visualization of omics data using g:Profiler, GSEA, Cytoscape and EnrichmentMap. Nat Protoc. 2019;14(2):482-517. doi: 10.1038/s41596-018-0103-9
- Kong J, Lee H, Kim D, et al. Network-based machine learning (ML) in colorectal and bladder organoid models predicts anti-cancer drug efficacy in patients. Nat Commun. 2020;11(1):5485. doi: 10.1038/s41467-020-19313-8
- Kong J, Ha D, Lee J, et al. Network-based machine learning (ML) approach to predict immunotherapy response in cancer patients. Nat Commun. 2022;13(1):3703. doi: 10.1038/s41467-022-31535-6
- Suleman M, Ali S, Rashid F, Khan FI. Experimental and computational methods in the development of diagnostics and therapeutics for colon cancer. Front Mol Biosci. 2025;12:1568721. doi: 10.3389/fmolb.2025.1568721
- Ivancic MM, Megna BW, Sverchkov Y, et al. Noninvasive detection of colorectal carcinomas using serum protein biomarkers. J Surg Res. 2020;246:160-169. doi: 10.1016/j.jss.2019.08.004
- Wu Z, Li Y, Zhang Y, et al. Colorectal cancer screening methods and molecular markers for early detection. Technol Cancer Res Treat. 2020;19:1533033820980426. doi: 10.1177/1533033820980426
- Dhillon A, Singh A, Bhalla VK. A systematic review on biomarker identification for cancer diagnosis and prognosis in multi-omics: from computational needs to machine learning (ML) and deep learning. Arch Comput Methods Eng. 2023;30(2):917-949. doi: 10.1007/s11831-022-09821-9
- Al-Tashi Q, Saad MB, Muneer A, et al. Machine learning (ML) models for the identification of prognostic and predictive cancer biomarkers: a systematic review. Int J Mol Sci. 2023;24(9):7781. doi: 10.3390/ijms24097781
- Skrede OJ, De Raedt S, Kleppe A, et al. Deep learning for prediction of colorectal cancer outcome: a discovery and validation study. Lancet. 2020;395(10221):350-360. doi: 10.1016/S0140-6736(19)32998-8
- Oh HH, Joo YE. Novel biomarkers for the diagnosis and prognosis of colorectal cancer. Intest Res. 2020;18(2):168- 183. doi: 10.5217/IR.2019.00080
- Rahman MR, Islam T, Gov E, et al. Identification of prognostic biomarker signatures and candidate drugs in colorectal cancer: insights from systems biology analysis. Medicina. 2019;55(1):20. doi: 10.3390/medicina55010020
- Pavitra E, Dariya B, Srivani G, et al. Engineered nanoparticles for imaging and drug delivery in colorectal cancer. Semin Cancer Biol. 2021;69:293-306. doi: 10.1016/j.semcancer.2019.06.017
- Krasteva N, Georgieva M. Promising therapeutic strategies for colorectal cancer treatment based on nanomaterials. Pharmaceutics. 2022;14(6):1213. doi: 10.3390/pharmaceutics14061213
- Carvalho MR, Reis RL, Oliveira JM. Dendrimer nanoparticles for colorectal cancer applications. J Mater Chem B. 2020;8(6):1128-1138. doi: 10.1039/C9TB02289A
- Brar B, Ranjan K, Palria A, et al. Nanotechnology in colorectal cancer for precision diagnosis and therapy. Front Nanotechnol. 2021;3:699266. doi: 10.3389/fnano.2021.699266
- Gogoi P, Kaur G, Singh NK. Nanotechnology for colorectal cancer detection and treatment. World J Gastroenterol. 2022;28(46):6497-6511. doi: 10.3748/wjg.v28.i46.6497
- Ghorbani F, Kokhaei P, Ghorbani M, Eslami M. Application of different nanoparticles in the diagnosis of colorectal cancer. Gene Rep. 2020;21:100896. doi: 10.1016/j.genrep.2020.100896
- Barabadi H, Vahidi H, Damavandi Kamali K, et al. Emerging theranostic gold nanomaterials to combat colorectal cancer: a systematic review. J Clust Sci. 2020;31(4):651-658. doi: 10.1007/s10876-019-01681-x
- Vairavel M, Devaraj E, Shanmugam R. An eco-friendly synthesis of Enterococcus sp.–mediated gold nanoparticle induces cytotoxicity in human colorectal cancer cells. Environ Sci Pollut Res Int. 2020;27(8):8166-8175. doi: 10.1007/s11356-019-07511-x
- Mármol I, Quero J, Rodríguez-Yoldi MJ, Cerrada E. Gold as a possible alternative to platinum-based chemotherapy for colon cancer treatment. Cancers. 2019;11(6):780. doi: 10.3390/cancers11060780
- Emami F, Banstola A, Vatanara A, et al. Doxorubicin and anti-PD-L1 antibody conjugated gold nanoparticles for colorectal cancer photochemotherapy. Mol Pharm. 2019;16(3):1184-1199. doi: 10.1021/acs.molpharmaceut.8b01157
- Lin D, Feng S, Pan J, et al. Colorectal cancer detection by gold nanoparticle based surface-enhanced Raman spectroscopy of blood serum and statistical analysis. Opt Express. 2011;19(14):13565-13577. doi: 10.1364/OE.19.013565
- Liu J, Zhan J, Zhang Y, et al. Ultrathin clay nanoparticles-mediated mutual reinforcement of ferroptosis and cancer immunotherapy. Adv Mater. 2024;36(9):e2309562. doi: 10.1002/adma.202309562
- Mokhosi SR, Mdlalose W, Nhlapo A, Singh M. Advances in the synthesis and application of magnetic ferrite nanoparticles for cancer therapy. Pharmaceutics. 2022;14(5):937. doi: 10.3390/pharmaceutics14050937
- Egwuche B, Aisida SO, Al Nasir MH, et al. Manganese-doped zinc oxide nanoparticles capped with chitosan, cetyltrimethylammonium bromide and Gongronema latifolium for hyperthermia applications. J Macromol Sci Part B. 2024:1-23. doi: 10.1080/00222348.2024.2425562
- Alfareed TM, Slimani Y, Almessiere MA, et al. Biocompatibility and colorectal anti-cancer activity study of nanosized BaTiO3 coated spinel ferrites. Sci Rep. 2022;12:14127. doi: 10.1038/s41598-022-18306-5
- Zhao Y, Xu J, Le VM, et al. EpCAM aptamer-functionalized cationic liposome-based nanoparticles loaded with miR- 139-5p for targeted therapy in colorectal cancer. Mol Pharm. 2019;16(11):4696-4710. doi: 10.1021/acs.molpharmaceut.9b00867
- Barani M, Bilal M, Rahdar A, et al. Nanodiagnosis and nanotreatment of colorectal cancer: an overview. J Nanopart Res. 2021;23(1):18. doi: 10.1007/s11051-020-05129-6
- Mansoori B, Mohammadi A, Abedi-Gaballu F, et al. Hyaluronic acid-decorated liposomal nanoparticles for targeted delivery of 5-fluorouracil into HT-29 colorectal cancer cells. J Cell Physiol. 2020;235(10):6817-6830. doi: 10.1002/jcp.29576
- Yang C, Merlin D. Lipid-based drug delivery nanoplatforms for colorectal cancer therapy. Nanomaterials. 2020;10(7):1424. doi: 10.3390/nano10071424
- Abrishami A, Bahrami AR, Nekooei S, Saljooghi AS, Matin MM. Hybridized quantum dot, silica, and gold nanoparticles for targeted chemo-radiotherapy in colorectal cancer theranostics. Commun Biol. 2024;7(1):393. doi: 10.1038/s42003-024-06043-6
- Khan FA, Albalawi R, Pottoo FH. Trends in targeted delivery of nanomaterials in colon cancer diagnosis and treatment. Med Res Rev. 2022;42(1):227-258. doi: 10.1002/med.21809
- Qureshi A, Tufani A, Corapcioglu G, Niazi JH. CdSe/CdS/ ZnS nanocrystals decorated with Fe3O4 nanoparticles for point-of-care optomagnetic detection of cancer biomarker in serum. Sens Actuators B Chem. 2020;321:128431. doi: 10.1016/j.snb.2020.128431
- Sharma P, Gupta R, Iqbal M, et al. Photothermally active quantum dots in cancer imaging and therapeutics: nanotheranostics perspective. ACS Appl Bio Mater. 2024;7(4):1987-2005. doi: 10.1021/acsabm.4c01190
- Guo Y, Wang M, Zou Y, et al. Mechanisms of chemotherapeutic resistance and the application of targeted nanoparticles for enhanced chemotherapy in colorectal cancer. J Nanobiotechnol. 2022;20(1):371. doi: 10.1186/s12951-022-01586-4
- Wahnou H, Liagre B, Sol V, et al. Polyphenol-based nanoparticles: a promising frontier for enhanced colorectal cancer treatment. Cancers. 2023;15(15):3826. doi: 10.3390/cancers15153826
- Choukaife H, Seyam S, Alallam B, Doolaanea AA, Alfatama M. Current advances in chitosan nanoparticles based oral drug delivery for colorectal cancer treatment. Int J Nanomed. 2022;17:3933-3966. doi: 10.2147/IJN.S375229
- Al-Joufi FA, Setia A, Salem-Bekhit MM, et al. Molecular pathogenesis of colorectal cancer with an emphasis on recent advances in biomarkers, as well as nanotechnology-based diagnostic and therapeutic approaches. Nanomaterials. 2022;12(1):169. doi: 10.3390/nano12010169
- Luo B, Zhou J, Li Z, et al. Ultrasensitive DNA methylation ratio detection based on the target-induced nanoparticle-coupling and site-specific base oxidation damage for colorectal cancer. Anal Chem. 2022;94(16):6261–6270. doi: 10.1021/acs.analchem.2c00104
- Perumal V, Sivakumar PM, Zarrabi A, et al. Near infra-red polymeric nanoparticle based optical imaging in cancer diagnosis. J Photochem Photobiol B. 2019;199:111630. doi: 10.1016/j.jphotobiol.2019.111630
- Girigoswami K, Girigoswami A. A review on the role of nanosensors in detecting cellular miRNA expression in colorectal cancer. Endocr Metab Immune Disord Drug Targets. 2021;21(1):12–26. doi: 10.2174/1871530320666200515115723
- Hanoglu SB, Man E, Harmanci D, et al. Magnetic nanoparticle-based electrochemical sensing platform using ferrocene-labelled peptide nucleic acid for the early diagnosis of colorectal cancer. Biosensors. 2022;12(9):736. doi: 10.3390/bios12090736
- Zhang X, Tan X, Wang P, Qin J. Application of polypyrrole-based electrochemical biosensor for the early diagnosis of colorectal cancer. Nanomaterials. 2023;13(4):674. doi: 10.3390/nano13040674
- Beniwal SS, Lamo P, Kaushik A, et al. Current status and emerging trends in colorectal cancer screening and diagnostics. Biosensors. 2023;13(10):926. doi: 10.3390/bios13100926
- Chow JC. Nanomaterial-based molecular imaging in cancer: advances in simulation and AI integration. Biomolecules. 2025;15(3):444. doi: 10.3390/biom15030444
- Pandurangan P, Rakshi AD, Sundar MSAV, et al. Integrating cutting-edge technologies: AI, IoT, blockchain and nanotechnology for enhanced diagnosis and treatment of colorectal cancer-A review. J Drug Deliv Sci Technol. 2024;91:105197. doi: 10.1016/j.jddst.2023.105197
- Vallejo Morales E, Suárez Guerrero G, Hoyos Palacio LM. Computational simulation of colorectal cancer biomarker particle mobility in a 3D model. Molecules. 2023;28(2):589. doi: 10.3390/molecules28020589
- Kamal S, Derbala HA, Alterary SS, et al. Synthesis, biological, and molecular docking studies on 4,5,6,7-tetrahydrobenzo[b] thiophene derivatives and their nanoparticles targeting colorectal cancer. ACS Omega. 2021;6(43):29263–29275. doi: 10.1021/acsomega.1c04063
- Al-Kabani A, Huda B, Haddad J, et al. Exploring experimental models of colorectal cancer: a critical appraisal from 2D cell systems to organoids, humanized mouse avatars, organ-on-chip, CRISPR engineering, and AI-driven platforms—challenges and opportunities for translational precision oncology. Cancers. 2025;17(13):2163. doi: 10.3390/cancers17132163
- Hani U, Mahammed N, Reshma T, et al. Enhanced colon-targeted drug delivery through development of 5-fluorouracil-loaded cross-linked mastic gum nanoparticles. Sci Rep. 2025;15:18355. doi: 10.1038/s41598-025-03533-3
- Djermane R, Nieto C, Vega MA, del Valle EMM. Antibody-loaded nanoplatforms for colorectal cancer diagnosis and treatment: an update. Pharmaceutics. 2023;15(5):1514. doi: 10.3390/pharmaceutics15051514
- Nie D, Zhu Y, Guo T, Yue M, Lin M. Research advance in manganese nanoparticles in cancer diagnosis and therapy. Front Mater. 2022;9:857385. doi: 10.3389/fmats.2022.857385
- Ma L, Wang X, Wu Y, et al. Controlled release of manganese and magnesium ions by microsphere-encapsulated hydrogel enhances cancer immunotherapy. J Control Release. 2024;372:682–698. doi: 10.1016/j.jconrel.2024.06.067
- Jain P, Patel K, Jangid AK, et al. Modulating the delivery of 5-fluorouracil to human colon cancer cells using multifunctional arginine-coated manganese oxide nanocuboids with MRI properties. ACS Appl Bio Mater. 2020;3(10):6852–6864. doi: 10.1021/acsabm.0c00780
- Foroushani MS, Shervedani RK, Kefayat A, et al. Folate-graphene chelate manganese nanoparticles as a theranostic system for colon cancer MR imaging and drug delivery: In-vivo examinations. J Drug Deliv Sci Technol. 2019;54:101223. doi: 10.1016/j.jddst.2019.101223
- Liang S, Liao G, Zhu W, Zhang L. Manganese-based hollow nanoplatforms for MR imaging-guided cancer therapies. Biomater Res. 2022;26(1):32. doi: 10.1186/s40824-022-00275-5
- Kourani K, Jain P, Kumar A, et al. Inulin coated Mn3O4 nanocuboids coupled with RNA interference reverse intestinal tumorigenesis in Apc knockout murine colon cancer models. Nanomedicine. 2022;40:102504. doi: 10.1016/j.nano.2021.102504
- Li H, Feng X, Li H, et al. The supplement of magnesium element to inhibit colorectal tumor cells. Biol Trace Elem Res. 2023;201(6):2895–2903. doi: 10.1007/s12011-022-03393-2
- Elhawary EA, Soltane R, Moustafa MH, et al. Sustainable MnO2/MgO bimetallic nanoparticles capped with sword fern methanol extract attain antioxidant/anti-biofilm potential: a UPLC-ESI/LC/MS and network pharmacology-supported study. Pharmaceuticals. 2025;18(9):1262. doi: 10.3390/ph18091262
- Mukherjee S, Madamsetty VS, Pradhan L, Upadhyay A, Yenurkar D. Magnetic Nanomaterials: Innovation for Cancer Theranostics. IOP Publishing; 2025. doi: 10.1088/978-0-7503-6335-8
- Eyube MO, Enuesueke C, Alimikhena M. Biocompatibility of nanomaterials in medical applications. Innov Med Omics. 2025;2(3):44–58. doi: 10.36922/IMO025210024
- Saddik MS, Elsayed MM, Abdelkader MSA, et al. Novel green biosynthesis of 5-fluorouracil chromium nanoparticles using Harpullia pendula extract for treatment of colorectal cancer. Pharmaceutics. 2021;13(2):226. doi: 10.3390/pharmaceutics13020226
- Althomali A, Daghestani MH, Almukaynizi FB, et al. Anti-colon cancer activities of green-synthesized Moringa oleifera–AgNPs against human colon cancer cells. Green Process Synth. 2022;11(1):545–554. doi: 10.1515/gps-2022-0052
- Jain A, Bhattacharya S. Recent advances in nanomedicine preparative methods and their therapeutic potential for colorectal cancer: a critical review. Front Oncol. 2023;13:1211603. doi: 10.3389/fonc.2023.1211603
- Koumpa FS, Xylas D, Konopka M, et al. Colorectal peritoneal metastases: a systematic review of current and emerging trends in clinical and translational research. Gastroenterol Res Pract. 2019;2019:5180895. doi: 10.1155/2019/5180895
- Kasi PB, Mallela VR, Ambrozkiewicz F, et al. Theranostics nanomedicine applications for colorectal cancer and metastasis: recent advances. Int J Mol Sci. 2023;24(9):7922. doi: 10.3390/ijms24097922
- Gong S, Jin X, Guo Y, Yu J. NLP for computational insights into nutritional impacts on colorectal cancer care. SLAS Technol. 2025;32:100295. doi: 10.1016/j.slast.2025.100295
- Nair AS, Mishra SK, Johny A, Chandran KP, George JJ. SARS, MERS, and COVID-19: knowledge mining through bioinformatics and computational biology. In: Emerging and Re-Emerging Viral Diseases. CRC Press; 2025:244–248. doi: 10.1201/9781032721811-17
- Guimarães PAS, Carvalho MGR, Ruiz JC. A computational framework for extracting biological insights from SRA cancer data. Sci Rep. 2025;15(1):8117. doi: 10.1038/s41598-025-91781-8
- Usifo OR, Isoje AO, Kolawole OM. Evaluation of the effects of Arachis hypogaea and 5-fluorouracil on biochemical and histological parameters in 1,2-dimethylhydrazine-induced colon carcinogenesis in mice. Bull Nat Appl Sci. 2025;2(2):139–147. doi: 10.5281/zenodo/15832235
- Limmongkon A, Bueschl C, Doppler M, et al. Molecular networking of arachidin-related compounds from elicited peanut hairy root culture and their potential activity against HCT116 colon cancer cells. Plant Cell Tissue Organ Cult. 2025;160(2):54. doi: 10.1007/s11240-025-02993-9
- Roy S, Deka D, Kondaveeti SB, et al. An overview of potential of natural compounds to regulate epigenetic modifications in colorectal cancer: a recent update. Epigenetics. 2025;20(1):2491316. doi: 10.1080/15592294.2025.2491316
- Zhou L, Zhang J, Zhao K, Chen B, Sun Z. Natural products modulating MAPK for CRC treatment: a promising strategy. Front Pharmacol. 2025;16:1514486. doi: 10.3389/fphar.2025.1514486
- Karati D, Mukherjee S, Basu B, et al. Unlocking the potential of RNA nanoparticles: a breakthrough approach to overcoming challenges in colon cancer treatment. Curr Pharm Biotechnol. 2025;26(7):992–1013. doi: 10.2174/0113892010285554240303160500
- Mandala G. Integrating multi-omics data and AI for early detection and personalized treatment of colorectal cancer. Am J Psychiatr Rehabil. 2025;28(1):73–91. doi: 10.69980/ajpr.v28i1.68
- Sun K, Wang Y, Qu R, et al. Comprehensive application of artificial intelligence in colorectal cancer: a review. iScience. 2025;26(7):112980. doi: 10.1016/j.isci.2025.112980
- Hassane M. Artificial intelligence-driven precision medicine in cancer treatment. Science. 2024;1:100041. doi: 10.70389/PJS.100041
- Mao Y, Shangguan D, Huang Q, et al. Emerging artificial intelligence-driven precision therapies in tumor drug resistance: recent advances, opportunities, and challenges. Mol Cancer. 2025;24(1):123. doi: 10.1186/s12943-025-02321-x
- Yadav P, Singh D, Verma SK, et al. Regulatory framework and standards for bio-based materials. In: Handbook of Bio- Based Materials for Smart Manufacturing. 1st ed. CRC Press; 2025:20. doi: 10.1201/9781003566205-5

