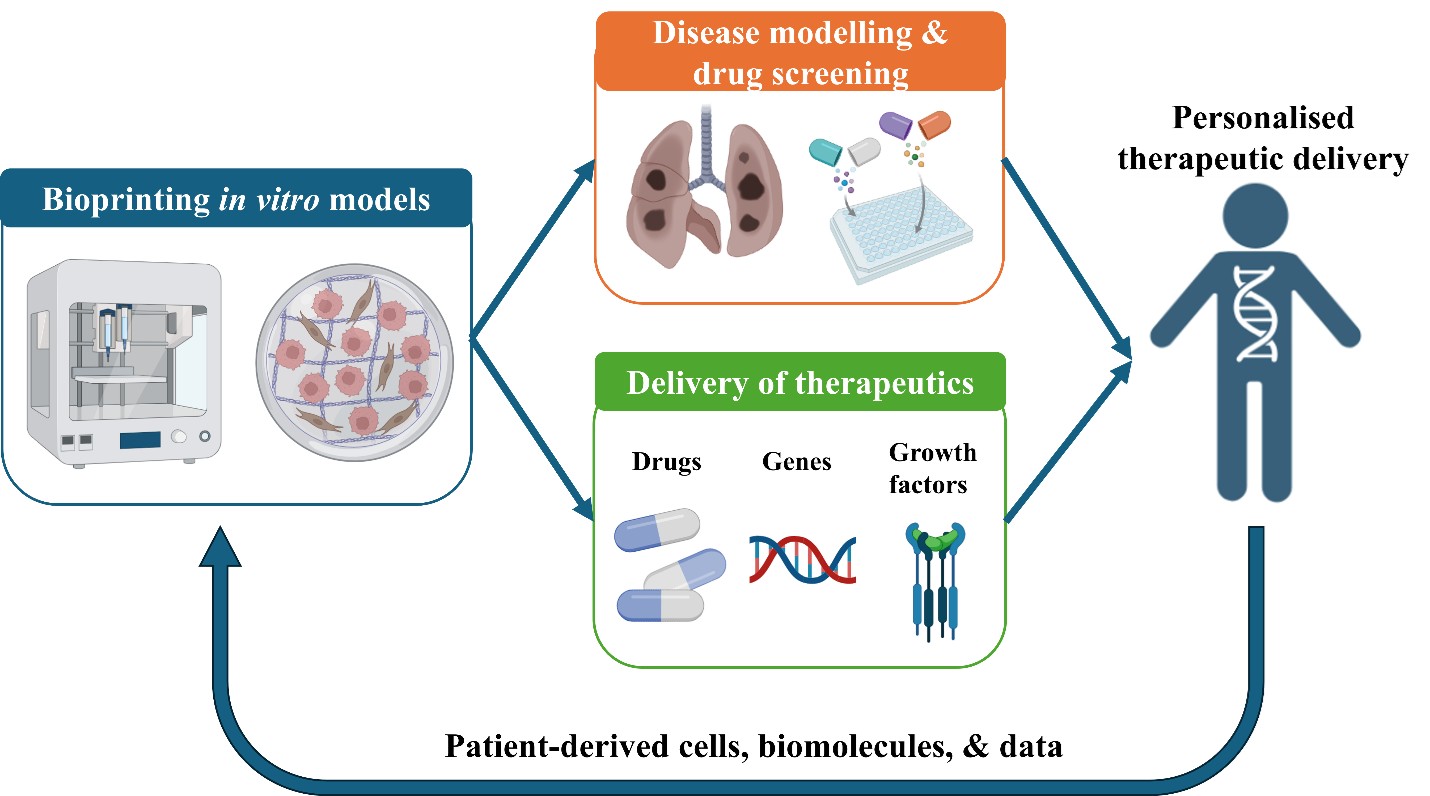
Bioprinting of in vitro models for personalized therapeutic delivery

Personalized therapeutic delivery aims to match the type, dose, timing, and localisation of treatment to each patient’s unique biological profile, requiring platforms that can model individual responses and precisely control how therapeutics are released. Achieving this precision is challenging because conventional 2D cultures and animal models fail to reproduce the 3D architecture and microenvironmental cues that shape drug, gene, and growth-factor dynamics in human tissues. Bioprinted in vitro models address these limitations by enabling the spatially defined assembly of cells, hydrogels, and bioactive components into physiologically relevant constructs. This review examines how bioprinting is advancing personalized therapeutic delivery, focusing on how bioink chemistry, construct architecture, and matrix mechanics influence transport behavior, release kinetics, and overall therapeutic performance. We highlight bioprinted liver, cardiac, and tumor models as predictive testbeds for evaluating patient-specific responses, and discuss advanced delivery strategies, including in situ bioprinting and 4D adaptive systems. Together, these developments position bioprinted in vitro platforms as integrated tools for designing, testing, and optimizing personalized therapeutic interventions within the broader framework of personalized medicine.
- Su J, Yang L, Sun Z, Zhan X. Personalized drug therapy: Innovative concept guided with proteoformics. Mol Cell Proteomics. 2024;23(3):100737. doi: 10.1016/j.mcpro.2024.100737
- Puccetti M, Pariano M, Schoubben A, Giovagnoli S, Ricci M. Biologics, theranostics, and personalized medicine in drug delivery systems. Pharmacol Res. 2024;201:107086. doi: 10.1016/j.phrs.2024.107086
- Wang D, Villenave R, Stokar-Regenscheit N, Clevers H. Human organoids as 3D in vitro platforms for drug discovery: Opportunities and challenges. Nat Rev Drug Discov. 2025;1-23. doi: 10.1038/s41573-025-01317-y
- Goetz LH, Schork NJ. Personalized medicine: Motivation, challenges, and progress. Fertil Steril. 2018;109(6):952-963. doi: 10.1016/j.fertnstert.2018.05.006
- Mani S, Lalani SR, Pammi M. Genomics and multiomics in the age of precision medicine. Pediatr Res. 2025;97(4):1399-1410. doi: 10.1038/s41390-025-04021-0
- Wang H, Xu X, Qin Y, et al. Wet-electrospun porous freeform scaffold enhances colonisation of cells. Mater Today Bio. 2025;33:101997. doi: 10.1016/j.mtbio.2025.101997
- Duval K, Grover H, Han LH, et al. Modeling physiological events in 2D vs. 3D cell culture. Physiology (Bathesda). 2017;32(4):266-277. doi: 10.1152/physiol.00036.2016
- Khetani SR, Bhatia SN. Microscale culture of human liver cells for drug development. Nat Biotechnol. 2008;26(1):120-126. doi: 10.1038/nbt1361
- Murphy SV, De Coppi P, Atala A. Opportunities and challenges of translational 3D bioprinting. Nat Biomed Eng. 2020;4(4):370-380. doi: 10.1038/s41551-019-0471-7
- Zhang YS, Haghiashtiani G, Hübscher T, et al. 3D extrusion bioprinting. Nat Rev Methods Primers. 2021;1(1):75. doi: 10.1038/s43586-021-00073-8
- Tripathi S, Dash M, Chakraborty R, et al. Engineering considerations in the design of tissue specific bioink for 3D bioprinting applications. Biomater Sci. 2025;13(1):93-129. doi: 10.1039/d4bm01192a
- Chen H, Cheng R, Chung SH, et al. Direct ink writing of bioactive PCL/laponite bone Implants: Engineering the interplay of design, process, structure, and function. Biomed Technol. 2025;11:100101. doi: 10.1016/j.bmt.2025.100101
- Abbadessa A, Ronca A, Salerno A. Integrating bioprinting, cell therapies and drug delivery towards in vivo regeneration of cartilage, bone and osteochondral tissue. Drug Deliv Transl Res. 2024;14(4):858-894. doi: 10.1007/s13346-023-01437-1
- Nie J, Gao Q, Fu J, He Y. Grafting of 3D bioprinting to in vitro drug screening: A review. Adv Healthc Mater. 2020;9(7):1901773. doi: 10.1002/adhm.201901773
- Peng W, Datta P, Ayan B, Ozbolat V, Sosnoski D, Ozbolat IT. 3D bioprinting for drug discovery and development in pharmaceutics. Acta Biomater. 2017;57:26-46. doi: 10.1016/j.actbio.2017.05.025
- Jain P, Kathuria H, Dubey N. Advances in 3D bioprinting of tissues/organs for regenerative medicine and in-vitro models. Biomaterials. 2022;287:121639. doi: 10.1016/j.biomaterials.2022.121639
- Mota C, Camarero-Espinosa S, Baker MB, Wieringa P, Moroni L. Bioprinting: From tissue and organ development to in vitro models. Chem Rev. 2020;120:10547-10607. doi: 10.1021/acs.chemrev.9b00789
- Mahmoudi Z, Sedighi M, Jafari A, et al. In situ 3D bioprinting: A promising technique in advanced biofabrication strategies. Bioprinting. 2023;31:e00260. doi: 10.1016/j.bprint.2023.e00260
- Samandari M, Mostafavi A, Quint J, Memić A, Tamayol A. In situ bioprinting: Intraoperative
implementation of regenerative medicine. Trends Biotechnol. 2022;40(10):1229-1247. doi: 10.1016/j.tibtech.2022.03.009
- Wan Z, Zhang P, Liu Y, Lv L, Zhou Y. Four-dimensional bioprinting: Current developments and applications in bone tissue engineering. Acta Biomater. 2020;101:26-42. doi: 10.1016/j.actbio.2019.10.038
- Gungor-Ozkerim PS, Inci I, Zhang YS, Khademhosseini A, Dokmeci MR. Bioinks for 3D bioprinting: An overview. Biomater Sci. 2018;6(5):915-946. doi: 10.1039/c7bm00765e
- Gopinathan J, Noh I. Recent trends in bioinks for 3D printing. Biomater Res. 2018;22(1):11. doi: 10.1186/s40824-018-0122-1
- Chen H, Hardwick J, Gao L, Plasencia DM, Subramanian S, Hirayama R. Acoustics in additive manufacturing: A path toward contactless, scalable, and high-precision manufacturing. Appl Phys Rev. 2025;12(3):031305. doi: 10.1063/5.0271688
- Shi Y, Tang S, Yuan X, et al. In situ 4D printing of polyelectrolyte/magnetic composites for sutureless gastric perforation sealing. Adv Mater. 2024;36(34):2307601. doi: 10.1002/adma.202307601
- Keriquel V, Oliveira H, Rémy M, et al. In situ printing of mesenchymal stromal cells, by laser-assisted bioprinting, for in vivo bone regeneration applications. Sci Rep. 2017;7(1):1778. doi: 10.1038/s41598-017-01914-x
- Ozbolat IT, Yu Y. Bioprinting toward organ fabrication: Challenges and future trends. IEEE Trans Biomed Eng. 2013;60(3):691-699. doi: 10.1109/TBME.2013.2243912
- Hopp B, Smausz T, Kresz N, et al. Survival and proliferative ability of various living cell types after laser-induced forward transfer. Tissue Eng. 2005;11(11-12):1817-1823. doi: 10.1089/ten.2005.11.1817
- Gruene M, Deiwick A, Koch L, et al. Laser printing of stem cells for biofabrication of scaffold-free autologous grafts. Tissue Eng Part C Methods. 2010;17(1):79-87. doi: 10.1089/ten.tec.2010.0359
- Koch L, Kuhn S, Sorg H, et al. Laser printing of skin cells and human stem cells. Tissue Eng Part C Methods. 2009;16(5):847-854. doi: 10.1089/ten.tec.2009.0397
- Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785. doi: 10.1038/nbt.2958
- Lee V, Dias A, Ozturk M, et al. 3D bioprinting and 3D imaging for stem cell engineering. In: Bioprinting in Regenerative Medicine. Berlin: ResearchGate; 2015. p. 33-66.
- Koo Y, Kim G. New strategy for enhancing in situ cell viability of cell-printing process via piezoelectric transducer-assisted three-dimensional printing. Biofabrication. 2016;8(2):025010. doi: 10.1088/1758-5090/8/2/025010
- Guillotin B, Souquet A, Catros S, et al. Laser assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials. 2010;31(28):7250-7256. doi: 10.1016/j.biomaterials.2010.05.055
- Barron JA, Wu P, Ladouceur HD, Ringeisen BR. Biological laser printing: A novel technique for creating heterogeneous 3-dimensional cell patterns. Biomed Microdev. 2004; 6(2):139-147. doi: 10.1023/B: BMMD.0000031751.67267.9f
- Schiele NR, Chrisey DB, Corr DT. Gelatin-based laser direct-write technique for the precise spatial patterning of cells. Tissue Eng Part C Methods. 2011;17(3):289-298. doi: 10.1089/ten.TEC.2010.0442
- Chen H, Zhang B, Huang J. Recent advances and applications of artificial intelligence in 3D bioprinting. Biophys Rev (Melville). 2024;5(3):031301. doi: 10.1063/5.0190208
- Foyt DA, Norman MDA, Yu TTL, Gentleman E. Exploiting advanced hydrogel technologies to address key challenges in regenerative medicine. Adv Healthc Mater. 2018;7(8):1700939. doi: 10.1002/adhm.201700939
- Pereira RF, Bártolo PJ. 3D bioprinting of photocrosslinkable hydrogel constructs. J Appl Polym Sci. 2015;132(48). doi: 10.1002/app.42458
- Hölzl K, Lin S, Tytgat L, Van Vlierberghe S, Gu L, Ovsianikov A. Bioink properties before, during and after 3D bioprinting. Biofabrication. 2016;8(3):032002. doi: 10.1088/1758-5090/8/3/032002
- Xu T, Kincaid H, Atala A, Yoo JJ. High-throughput production of single-cell microparticles using an inkjet printing technology. J Manuf Sci Eng Trans ASME. 2008;130(2):0210171-0210175. doi: 10.1115/1.2903064
- Cui X, Boland T, D’Lima DD, Lotz MK. Thermal inkjet printing in tissue engineering and regenerative medicine. Recent Pat Drug Deliv Formul. 2012;6(2):149-155. doi: 10.2174/187221112800672949
- Chen H, Khong J, Huang J. Direct ink writing of polycaprolactone/laponite composite for bone implants: 3D characterization using x-ray micro CT. Orthop Proc. 2021;103-B(SUPP_16):74-74.
- Jones N. Science in three dimensions: The print revolution. Nature. 2012;487(7405):22-23. doi: 10.1038/487022a
- Pati F, Jang J, Lee JW, Cho DW. Extrusion bioprinting. In: Atala A, Yoo JJ, editors. Essentials of 3D Biofabrication and Translation. Ch. 7. United States: Academic Press; 2015. p. 123-152.
- Melchels FPW, Domingos MAN, Klein TJ, Malda J, Bartolo PJ, Hutmacher DW. Additive manufacturing of tissues and organs. Prog Polym Sci. 2012;37(8):1079-1104. doi: 10.1016/j.progpolymsci.2011.11.007
- Chen H, Stampoultzis T, Papadopoulou A, Balabani S, Huang J. Evaluation of rheological properties and shape fidelity of polycaprolactone/hydroxyapatite inks for 3D printing of osteochondral tissue scaffolds. Orthop Proceed. 2021;103-B(Supp 2):96.
- Ozbolat IT, Hospodiuk M. Current advances and future perspectives in extrusion-based bioprinting. Biomaterials. 2016;76:321-343. doi: 10.1016/j.biomaterials.2015.10.076
- Li W, Wang M, Ma H, Chapa-Villarreal FA, Lobo AO, Zhang YS. Stereolithography apparatus and digital light processing-based 3D bioprinting for tissue fabrication. Iscience. 2023;26(2):106039. doi: 10.1016/j.isci.2023.106039
- Kumar H, Kim K. Stereolithography 3D Bioprinting. 3D Bioprinting: Principles and Protocols. Berlin: Springer; 2020. p. 93-108.
- Guida L, Cavallaro M, Levi M. Advancements in high-resolution 3D bioprinting: Exploring technological trends, bioinks and achieved resolutions. Bioprinting. 2024;44:e00376. doi: 10.1016/j.bprint.2024.e00376
- Bao Y, Paunović N, Leroux JC. Challenges and opportunities in 3D printing of biodegradable medical devices by emerging photopolymerization techniques. Adv Funct Mater. 2022;32(15):2109864. doi: 10.1002/adfm.202109864
- Zhu Y, Guo S, Ravichandran D, et al. 3Dlprinted polymeric biomaterials for health applications. Adv Healthc Mater. 2025;14(1):2402571. doi: 10.1002/adhm.202402571
- Soliman BG, Longoni A, Major GS, et al. Harnessing macromolecular chemistry to design hydrogel micro‐and macro‐environments. Macromol Biosci. 2024;24(5):2300457. doi: 10.1002/mabi.202300457
- Cornelissen DJ, Faulkner-Jones A, Shu W. Current developments in 3D bioprinting for tissue engineering. Curr Opin Biomed Eng. 2017;2:76-82. doi: 10.1016/j.cobme.2017.05.004
- Slaughter BV, Khurshid SS, Fisher OZ, Khademhosseini A, Peppas NA. Hydrogels in regenerative medicine. Adv Mater. 2009;21(32(32;21(32 medi doi: 10.1002/adma.200802106
- Choi B, Kim S, Lin B, Wu BM, Lee M. Cartilaginous extracellular matrix-modified chitosan hydrogels for cartilage tissue engineering. ACS Appl Mater Interfaces. 2014;6(22):20110-20121. doi: 10.1021/am505723k
- Gibas I, Janik H. Review: Synthetic polymer hydrogels for biomedical applications. Chem Chem Technol. 2010;4:297-304. doi: 10.23939/chcht04.04.297
- Yazdimamaghani M, Vashaee D, Assefa S, et al. Hybrid macroporous gelatin/bioactive-glass/nanosilver scaffolds with controlled degradation behavior and antimicrobial activity for bone tissue engineering. J Biomed Nanotechnol. 2014;10:911-931. doi: 10.1166/jbn.2014.1783
- Van Vlierberghe S, Dubruel P, Schacht E. Biopolymer-based hydrogels as scaffolds for tissue engineering applications: A review. Biomacromolecules. 2011;12(5):1387-1408. doi: 10.1021/bm200083n
- Malda J, Visser J, Melchels FP, et al. 25th Anniversary article: Engineering hydrogels for biofabrication. Adv Mater. 2013;25(36):5011-5028. doi: 10.1002/adma.201302042
- Merceron TK, Murphy SV. Hydrogels for 3D bioprinting applications. In: Atala A, Yoo JJ, editors. Essentials of 3D Biofabrication and Translation. Ch. 14. United States: Academic Press; 2015. p. 249-270.
- Chirani N, Yahia LH, Gritsch L, Motta F, Chirani S, Farè S. History and applications of hydrogels. J Biomed Sci. 2015;4:13-23. doi: 10.4172/2254-609X.100013
- Yang D, Li L, Chen H, Wang C, Huang J, Zheng T. A rapid and bidirectional humidity-responsive actuator via one-step self-assembly of a PVDF@F127-TiO₂ monolithic membrane for smart wearables. Chem Eng J. 2025;519:165568. doi: 10.1016/j.cej.2025.165568
- Drury JL, Mooney DJ. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials. 2003;24(24):4337-4351. doi: 10.1016/S0142-9612(03)00340-5
- Li C, Zhou Z, Meng X, et al. A preliminary study on the “hitchhiking” of radionuclides on microplastics: A new threat to the marine environment from compound pollution. Toxics. 2025;13(6):429. doi: 10.3390/toxics13060429
- Critchley S, Kelly D. Bioinks for bioprinting functional meniscus and articular cartilage. J 3D Print Med. 2017;1:269-290. doi: 10.2217/3dp-2017-0012
- Rocha LB, Goissis G, Rossi MA. Biocompatibility of anionic collagen matrix as scaffold for bone healing. Biomaterials. 2002;23(2):449-456. doi: 10.1016/S0142-9612(01)00126-0
- Lee KY, Mooney DJ. Hydrogels for tissue engineering. Chem Rev. 2001;101(7):1869-1880. doi: 10.1021/cr000108x
- Panwar A, Tan LP. Current status of bioinks for micro-extrusion-based 3D bioprinting. Molecules. 2016;21(6):685. doi: 10.3390/molecules21060685
- Chen H. 3D Printing Hybrid Scaffold with Hydrogel and Filler-Loaded PCL for Osteochondral Tissue Engineering. London: UCL (University College London); 2023.
- Daly AC, Critchley SE, Rencsok EM, Kelly DJ. A comparison of different bioinks for 3D bioprinting of fibrocartilage and hyaline cartilage. Biofabrication. 2016;8(4):045002. doi: 10.1088/1758-5090/8/4/045002
- Jia J, Richards DJ, Pollard S, et al. Engineering alginate as bioink for bioprinting. Acta Biomaterialia. 2014;10(10):4323-4331. doi: 10.1016/j.actbio.2014.06.034
- Khalil S, Sun W. Bioprinting endothelial cells with alginate for 3D tissue constructs. J Biomech Eng. 2009;131(11):111002. doi: 10.1115/1.3128729
- Unagolla JM, Jayasuriya AC. Hydrogel-based 3D bioprinting: A comprehensive review on cell-laden hydrogels, bioink formulations, and future perspectives. Appl Mater Today. 2020;18:100479. doi: 10.1016/j.apmt.2019.100479
- Raucci MG, D’Amora U, Ronca A, Demitri C, Ambrosio L. Bioactivation routes of gelatin-based scaffolds to enhance at nanoscale level bone tissue regeneration. Front Bioeng Biotechnol. 2019;7:27. doi: 10.3389/fbioe.2019.00027
- Laronda MM, Rutz AL, Xiao S, et al. A bioprosthetic ovary created using 3D printed microporous scaffolds restores ovarian function in sterilized mice. Nat Commun. 2017;8(1):15261. doi: 10.1038/ncomms15261
- Chung JHY, Naficy S, Yue Z, et al. Bio-ink properties and printability for extrusion printing living cells. 10.1039/ C3BM00012E. Biomater Sci. 2013;1(7):763-773. doi: 10.1039/C3BM00012E
- Klotz BJ, Gawlitta D, Rosenberg AJWP, Malda J, Melchels FPW. Gelatin-methacryloyl hydrogels: Towards biofabrication-based tissue repair. Trends Biotechnol. 2016;34(5):394-407. doi: 10.1016/j.tibtech.2016.01.002
- Nguyen AH, McKinney J, Miller T, Bongiorno T, McDevitt TC. Gelatin methacrylate microspheres for controlled growth factor release. Acta Biomater. 2015;13:101-110. doi: 10.1016/j.actbio.2014.11.028
- Montazerian H, Baidya A, Haghniaz R, et al. Stretchable and bioadhesive gelatin methacryloyl-based hydrogels enabled by in Situ dopamine polymerization. ACS Appl Mater Interfaces. 2021;13(34):40290-40301. doi: 10.1021/acsami.1c10048
- Van Den Bulcke AI, Bogdanov B, De Rooze N, Schacht EH, Cornelissen M, Berghmans H. Structural and rheological properties of methacrylamide modified gelatin hydrogels. Biomacromolecules. 2000;1(1):31-38. doi: 10.1021/bm990017d
- Etale A, Onyianta AJ, Turner SR, Eichhorn SJ. Cellulose: A review of water interactions, applications in composites, and water treatment. Chem Rev. 2023;123(5):2016-2048. doi: 10.1021/acs.chemrev.2c00477
- Wang M, Jiang G, Guo X, Zeng S, Zhao D. Cellulose functional gels: Physical design and promising applications. Adv Phys Res. 2025;4(6):2500020. doi: 10.1002/apxr.202500020
- Sharkawy A, Barreiro MF, Rodrigues AE. Chitosan-based Pickering emulsions and their applications: A review. Carbohydr Polym. 2020;250:116885. doi: 10.1016/j.carbpol.2020.116885
- Yao Z, Feng X, Wang Z, et al. Techniques and applications in 3D bioprinting with chitosan bio-inks for drug delivery: A review. Int J Biol Macromol. 2024;278:134752. doi: 10.1016/j.ijbiomac.2024.134752
- Hacker M, Mikos AG. Synthetic polymers. In: Principles of Regenerative Medicine. San Diego: Academic Press; 2011. p. 587-622.
- Cui X, Breitenkamp K, Finn MG, Lotz M, D’Lima DD. Direct human cartilage repair using three-dimensional bioprinting technology. Tissue Eng Part A. 2012;18(11-12):1304-1312. doi: 10.1089/ten.TEA.2011.0543
- Gioffredi E, Boffito M, Calzone S, et al. Pluronic F127 hydrogel characterization and biofabrication in cellularized constructs for tissue engineering applications. Procedia CIRP. 2016;49:125-132. doi: 10.1016/j.procir.2015.11.001
- Nommeots-Nomm A, Lee PD, Jones JR. Direct ink writing of highly bioactive glasses. J Eur Ceram Soc. 2018;38(3):837-844. doi: 10.1016/j.jeurceramsoc.2017.08.006
- Diniz IMA, Chen C, Xu X, et al. Pluronic F-127 hydrogel as a promising scaffold for encapsulation of dental-derived mesenchymal stem cells. J Mater Sci. 2015;26(3):153. doi: 10.1007/s10856-015-5493-4
- Pepić I, Lovrić J, Hafner A, Filipović-Grčić J. Powder form and stability of Pluronic mixed micelle dispersions for drug delivery applications. Drug Dev Ind Pharm. 2014;40(7):944-951. doi: 10.3109/03639045.2013.791831
- Liu Y, Fu S, Lin L, et al. Redox-sensitive Pluronic F127- tocopherol micelles: Synthesis, characterization, and cytotoxicity evaluation. Int J Nanomedicine. 2017;12:2635-2644. doi: 10.2147/IJN.S122746
- Bearat HH, Vernon BL. Environmentally responsive injectable materials. In: Vernon B, editor. Injectable Biomaterials. Ch. 11. Delhi: Woodhead Publishing; 2011. p. 263-297.
- Kolesky DB, Truby RL, Gladman AS, Busbee TA, Homan KA, Lewis JA. 3D bioprinting of vascularized, heterogeneous cell-laden tissue constructs. Adv Mater. 2014;26(19):3124-3130. doi: 10.1002/adma.201305506
- Chang CC, Boland ED, Williams SK, Hoying JB. Direct-write bioprinting three-dimensional biohybrid systems for future regenerative therapies. J Biomed Mater Res Part B Appl Biomater. 2011;98B(1):160-170. doi: 10.1002/jbm.b.31831
- Samavedi S, Poindexter LK, Van Dyke M, Goldstein AS. Synthetic biomaterials for regenerative medicine applications. In: Orlando G, Lerut J, Soker S, Stratta RJ, editors. Regenerative Medicine Applications in Organ Transplantation. Ch. 7. United States: Academic Press; 2014. p. 81-99.
- Kobayashi M, Hyu HS. Development and evaluation of polyvinyl alcohol-hydrogels as an artificial atrticular cartilage for orthopedic implants. Materials. 2010;3(4):2753-2771. doi: 10.3390/ma3042753
- Muppalaneni S. Polyvinyl alcohol in medicine and pharmacy: A perspective. J Dev Drugs. 2013;02:3. doi: 10.4172/2329-6631.1000112
- Mandrycky C, Wang Z, Kim K, Kim DH. 3D bioprinting for engineering complex tissues. Biotechnol Adv. 2016;34(4):422-434. doi: 10.1016/j.biotechadv.2015.12.011
- Meng F, Meyer CM, Joung D, Vallera DA, McAlpine MC, Panoskaltsis-Mortari A. 3D bioprinted in vitro metastatic models via reconstruction of tumor microenvironments. Adv Mater. 2019;31(10):1806899. doi: 10.1002/adma.201806899
- Albritton JL, Miller JS. 3D bioprinting: Improving in vitro models of metastasis with heterogeneous tumor microenvironments. Dis Models Mech. 2017;10(1):3-14. doi: 10.1242/dmm.025049
- Unger C, Kramer N, Walzl A, Scherzer M, Hengstschläger M, Dolznig H. Modeling human carcinomas: Physiologically relevant 3D models to improve anti-cancer drug development. Adv Drug Deliv Rev. 2014;79-80:50-67. doi: 10.1016/j.addr.2014.10.015
- Gospodinova A, Nankov V, Tomov S, Redzheb M, Petrov PD. Extrusion bioprinting of hydroxyethylcellulose-based bioink for cervical tumor model. Carbohydr Polym. 2021;260:117793. doi: 10.1016/j.carbpol.2021.117793
- Cadena IA, Adhikari G, Almer A, et al. Development of a 3D in vitro human-sized model of cervical dysplasia to evaluate the delivery of ethyl cellulose-ethanol injection. Front Biomater Sci. 2024;3:1365781. doi: 10.3389/fbiom.2024.1365781
- Kizawa H, Nagao E, Shimamura M, Zhang G, Torii H. Scaffold-free 3D bio-printed human liver tissue stably maintains metabolic functions useful for drug discovery. Biochem Biophys Rep. 2017;10:186-191. doi: 10.1016/j.bbrep.2017.04.004
- Mittal N, Li H, Ananthanarayanan A, Yu H. Complex Interplay between Serum and Fibroblasts in 3D Hepatocyte Co-Culture. bioRxiv [Preprint]; 2018. p. 286088.
- Saberian E, Jenča A, Zafari Y, et al. Scaffold application for bone regeneration with stem cells in dentistry: Literature review. Cells. 2024;13(12):1065.
- Ferlin KM, Prendergast ME, Miller ML, Kaplan DS, Fisher JP. Influence of 3D printed porous architecture on mesenchymal stem cell enrichment and differentiation. Acta Biomater. 2016;32:161-169. doi: 10.1016/j.actbio.2016.01.007
- Ng WL, Goh MH, Yeong WY, Naing MW. Applying macromolecular crowding to 3D bioprinting: Fabrication of 3D hierarchical porous collagen-based hydrogel constructs. 10.1039/C7BM01015J. Biomater Sci. 2018;6(3):562-574. doi: 10.1039/C7BM01015J
- Mobaraki M, Ghaffari M, Yazdanpanah A, Luo Y, Mills DK. Bioinks and bioprinting: A focused review. Bioprinting. 2020;18:e00080. doi: 10.1016/j.bprint.2020.e00080
- Yu K, Yao Y, Gao Q, et al. Investigation of humidity-driven swelling- shrinking behavior of filaments in material extrusion of medical-grade biodegradable hydrogel. Int J Bioprint. 2025;11(4):409-25. doi: 10.36922/ijb025220222
- Shokrollahi P, Garg P, Wulff D, Hui A, Phan CM, Jones L. Vat photopolymerization 3D printing optimization: Analysis of print conditions and print quality for complex geometries and ocular applications. Int J Pharm. 2025;668:124999. doi: 10.1016/j.ijpharm.2024.124999
- Zennifer A, Subramanian A, Sethuraman S. Design considerations of bioinks for laser bioprinting technique towards tissue regenerative applications. Bioprinting. 2022;27:e00205. doi: 10.1016/j.bprint.2022.e00205
- Sheybanikashani S, Zandi N, Hosseini D, Lotfi R, Simchi A. A sustainable and self-healable silk fibroin nanocomposite with antibacterial and drug eluting properties for 3D printed wound dressings. J Mater Chem B. 2024;12(3):784-799. doi: 10.1039/D3TB02363J
- Jia Z, Xu X, Zhu D, Zheng Y. Design, printing, and engineering of regenerative biomaterials for personalized bone healthcare. Prog Mater Sci. 2023;134:101072. doi: 10.1016/j.pmatsci.2023.101072
- Gunawan B, Kaplowitz N. Clinical perspectives on xenobiotic-induced hepatotoxicity. Drug Metab Rev. 2004;36(2):301-312. doi: 10.1081/dmr-120034148
- Kang K, Kim Y, Jeon H, et al. Three-dimensional bioprinting of hepatic structures with directly converted hepatocyte-like cells. Tissue Eng Part A. 2018;24(7-8):576-583. doi: 10.1089/ten.TEA.2017.0161
- Zhang YS, Arneri A, Bersini S, et al. Bioprinting 3D microfibrous scaffolds for engineering endothelialized myocardium and heart-on-a-chip. Biomaterials. 2016;110:45-59. doi: 10.1016/j.biomaterials.2016.09.003
- Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics--2015 update: A report from the American Heart Association. Circulation. 2015;131(4):e329-322. doi: 10.1161/cir.0000000000000152
- Shi X, Cheng Y, Wang J, et al. 3D printed intelligent scaffold prevents recurrence and distal metastasis of breast cancer. Theranostics. 2020;10(23):10652-10664. doi: 10.7150/thno.47933
- Goyanes A, Kobayashi M, Martínez-Pacheco R, Gaisford S, Basit AW. Fused-filament 3D printing of drug products: Microstructure analysis and drug release characteristics of PVA-based caplets. Int J Pharm. 2016;514(1):290-295. doi: 10.1016/j.ijpharm.2016.06.021
- Madorran E, Stožer A, Bevc S, Maver U. In vitro toxicity model: Upgrades to bridge the gap between preclinical and clinical research. Bosn J Basic Med Sci. 2020;20(2):157-168. doi: 10.17305/bjbms.2019.4378
- Zhao Y, Yao R, Ouyang L, et al. Three-dimensional printing of Hela cells for cervical tumor model in vitro. Biofabrication. 2014;6(3):035001. doi: 10.1088/1758-5082/6/3/035001
- Park JY, Choi JC, Shim JH, et al. A comparative study on collagen type I and hyaluronic acid dependent cell behavior for osteochondral tissue bioprinting. Biofabrication. 2014;6(3):035004. doi: 10.1088/1758-5082/6/3/035004
- Singhvi G, Singh M. Review: In-vitro drug release characterization models. Int J Pharm Stud Res. 2011;2(1):77-84.
- Liu X, Zhao K, Gong T, et al. Delivery of growth factors using a smart porous nanocomposite scaffold to repair a mandibular bone defect. Biomacromolecules. 2014;15(3):1019-1030. doi: 10.1021/bm401911p
- Water JJ, Bohr A, Boetker J, et al. Three-dimensional printing of drug-eluting implants: Preparation of an antimicrobial polylactide feedstock material. J Pharm Sci. 2015;104(3):1099-1107. doi: 10.1002/jps.24305
- Jaffredo M, Duchamp O, Touya N, et al. Proof of concept of intracochlear drug administration by laser-assisted bioprinting in mice. Hear Res. 2023;438:108880. doi: 10.1016/j.heares.2023.108880
- Khvorostina MA, Mironov AV, Nedorubova IA, et al. 3D printed gene-activated sodium alginate hydrogel scaffolds. Gels. 2022;8(7):421. doi: 10.3390/gels8070421
- Jeon O, Park H, Leach JK, Alsberg E. Biofabrication of engineered tissues by 3D bioprinting of tissue specific high cell-density bioinks. Mater Today. 2025;86:172-182. doi: 10.1016/j.mattod.2025.03.021
- Cevher E, Sezer AD, Çağlar E. Gene Delivery Systems: Recent Progress in Viral and Non-Viral Therapy. London: InTechOpen; 2012. p. 500.
- Xu T, Rohozinski J, Zhao W, Moorefield EC, Atala A, Yoo JJ. Inkjet-mediated gene transfection into living cells combined with targeted delivery. Tissue Eng Part A. 2009;15(1):95-101. doi: 10.1089/ten.tea.2008.0095
- Kumar SR, Markusic DM, Biswas M, High KA, Herzog RW Clinical development of gene therapy: Results and lessons from recent successes. Mol Ther Methods Clin Dev. 2016;3:16034. doi: 10.1038/mtm.2016.34
- Kay MA. State-of-the-art gene-based therapies: The road ahead. Nat Rev Genet. 2011;12(5):316-328. doi: 10.1038/nrg2971
- Gutierrez L, Cauchon NS, Christian TR, Giffin MJ, Abernathy MJ. The confluence of innovation in therapeutics and regulation: Recent CMC considerations. J Pharm Sci. 2020;109(12):3524-3534. doi: 10.1016/j.xphs.2020.09.025
- Ozbolat IT, Peng W, Ozbolat V. Application areas of 3D bioprinting. Drug Discov Today. 2016;21(8):1257-1271. doi: 10.1016/j.drudis.2016.04.006
- Duvall CL, Prokop A, Gersbach CA, Davidson JM. Gene delivery into cells and tissues. In: Lanza R, Langer R, Vacanti J, editors. Principles of Tissue Engineering. Ch. 35. 4th ed. United States: Academic Press; 2014. p. 687-723.
- Xiang Y, Zhong Z, Yao EJ, Kiratitanaporn W, Suy MT, Chen S. 3D bioprinting of gene delivery scaffolds with controlled release. Bioprinting. 2023;31:e00270. doi: 10.1016/j.bprint.2023.e00270
- Tayalia P, Mooney DJ. Controlled growth factor delivery for tissue engineering. Adv Mater. 2009;21(32-33):3269-3285. doi: 10.1002/adma.200900241
- Lee K, Silva EA, Mooney DJ. Growth factor delivery-based tissue engineering: General approaches and a review of recent developments. J R Soc Interface. 2010;8(55):153-170. doi: 10.1098/rsif.2010.0223
- Freeman FE, Pitacco P, Van Dommelen LHA, et al. 3D bioprinting spatiotemporally defined patterns of growth factors to tightly control tissue regeneration. Sci Adv. 2020;6(33):eabb5093. doi: 10.1126/sciadv.abb5093
- Ennett AB, Kaigler D, Mooney DJ. Temporally regulated delivery of VEGF in vitro and in vivo. J Biomed Mater Res A. 2006;79A(1):176-184. doi: 10.1002/jbm.a.30771
- Guan J, Stankus JJ, Wagner WR. Biodegradable elastomeric scaffolds with basic fibroblast growth factor release. J Control Release. 2007;120(1-2):70-78. doi: 10.1016/j.jconrel.2007.04.002
- Freeman FE, Kelly DJ. Tuning alginate bioink stiffness and composition for controlled growth factor delivery and to spatially direct MSC fate within bioprinted tissues. Sci Rep. 2017;7(1):17042. doi: 10.1038/s41598-017-17286-1
- Markstedt K, Mantas A, Tournier I, Martínez Ávila H, Hägg D, Gatenholm P. 3D bioprinting human chondrocytes with nanocellulose-alginate bioink for cartilage tissue engineering applications. Biomacromolecules. 2015;16(5):1489-1496. doi: 10.1021/acs.biomac.5b00188
- Noor N, Shapira A, Edri R, Gal I, Wertheim L, Dvir T. 3D printing of personalized thick and perfusable cardiac patches and hearts. Adv Sci. 2019;6(11):1900344. doi: 10.1002/advs.201900344
- Singh S, Choudhury D, Yu F, Mironov V, Naing MW. In situ bioprinting - Bioprinting from benchside to bedside? Acta Biomater. 2020;101:14-25. doi: 10.1016/j.actbio.2019.08.045
- Jeong SH, Kim J, Thibault BC, et al. Intelligent In situ printing of multimaterial bioinks for first-aid wound care guided by eye-in-hand robot technology. Adv Mater Technol. 2024;9:2400060. doi: 10.1002/admt.202400060
- Chen H, Huang J. Artificial intelligence in advancing sustainability in bioprinting. IJB. 2025;11(4):133-153. doi: 10.36922/ijb025170164
- Chen H, Bansal S, Martinez Plasencia D, Huang J, Subramanian S, Hirayama R. Acoustophoretic in situ 3D Fabrication of Multi-Material and Porous Structures. Berlin: ResearchGate; 2024.
- Chen H, Bansal S, Plasencia DM, et al. Omnidirectional and multi-material In Situ 3D printing using acoustic levitation. Adv Mater Technol. 2025;10(9):2401792. doi: 10.1002/admt.202401792
- Sadraei A, Naghib SM. 4D printing of physical stimuli-responsive hydrogels for localized drug delivery and tissue engineering. Polymer Rev. 2025;65(1):104-168.
- Faber L, Yau A, Chen Y. Translational biomaterials of four-dimensional bioprinting for tissue regeneration. Biofabrication. 2024;16(1):012001. doi: 10.1088/1758-5090/acfdd0
- Zhang X, Liu C, Wang S, et al. Assessment of the mechanical and functional properties of nitinol alloys fabricated by laser powder bed fusion: Effect of strain rates. Mater Sci Eng A. 2024;916:147358. doi: 10.1016/j.msea.2024.147358
- Zhang X, Chang T, Chen H, et al. Optimizing laser parameters and exploring building direction dependence of corrosion behavior in NiTi alloys fabricated by laser powder bed fusion. J Mater Res Technol. 2024;33:4023-4032. doi: 10.1016/j.jmrt.2024.10.105
- Rafiee M, Farahani RD, Therriault D. Multi-material 3D and 4D printing: A survey. Adv Sci (Weinh). 2020;7(12):1902307. doi: 10.1002/advs.201902307
- Yang Q, Gao B, Xu F. Recent advances in 4D bioprinting. Biotechnol J. 2020;15(1):1900086. doi: 10.1002/biot.201900086
- Zhang H, Koens L, Lauga E, Mourran A, Möller M. A light-driven microgel rotor. Small. 2019;15(46):e1903379. doi: 10.1002/smll.201903379
- Cheng T, Tahouni Y, Sahin ES, et al. Weather-responsive adaptive shading through biobased and bioinspired hygromorphic 4D-printing. Nat Commun. 2024;15(1):10366. doi: 10.1038/s41467-024-54808-8
- Pal V, Gupta D, Liu S, et al. Interparticle crosslinked Ion24-54808-8e microgels for 3D and 4D (Bio) printing applications. Small. 2025;21:e02262. doi: 10.1002/smll.202502262
- Lukin I, Musquiz S, Erezuma I, et al. Can 4D bioprinting revolutionize drug development? Expert Opin Drug Discov. 2019;14(10):953-956. doi: 10.1080/17460441.2019.1636781
- Braun NJ, Galaska RM, Jewett ME, Krupa KA. Implementation of a dynamic co-culture model abated silver nanoparticle interactions and nanotoxicological outcomes in vitro. Nanomaterials (Basel). 2021;11(7):1807. doi: 10.3390/nano11071807
- Li YC, Zhang YS, Akpek A, Shin SR, Khademhosseini A. 4D bioprinting: The next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication. 2016;9(1):012001. doi: 10.1088/1758-5090/9/1/012001
- Villar G, Heron AJ, Bayley H. Formation of droplet networks that function in aqueous environments. Nat Nanotechnol. 2011;6(12):803-808. doi: 10.1038/nnano.2011.183
- Zhang Y, Hsu LHH, Jiang X. Living electronics. Nano Res. 2020;13(5):1205-1213. doi: 10.1007/s12274-019-2570-x
- Stacey E, Maddock M, Dottori M, Beirne S, Yue Z. Colinear extrusion as an alternative multi-material 3d bioprinting approach for tissue engineering applications. Biomed Mater Dev. 2025;1-18. doi: 10.1007/s44174-025-00560-6
- Yu K, Gao Q, Mi Y, et al. Computational investigation of a 3D-printed osteochondral interface scaffold with comprehensive interfacial mechanical properties. Int J Bioprint. 2025;11(2):8777. doi: 10.36922/ijb.8577
- Puistola P, Miettinen S, Skottman H, Mörö A. Novel strategy for multi-material 3D bioprinting of human stem cell based corneal stroma with heterogenous design. Mater Today Bio. 2024;24:100924. doi: 10.1016/j.mtbio.2023.100924
- Zhang Z, Zhou X, Fang Y, Xiong Z, Zhang T. AI-driven 3D bioprinting for regenerative medicine: From bench to bedside. Bioact Mater. 2025;45:201-230. doi: 10.1016/j.bioactmat.2024.11.021
- Ou KL, Hosseinkhani H. Development of 3D in vitro technology for medical applications. Int J Mol Sci. 2014;15(10):17938-17962. doi: 10.3390/ijms151017938
- Malekpour A, Chen X. Printability and cell viability in extrusion-based bioprinting from experimental, computational, and machine learning views. J Funct Biomater. 2022;13(2):40. doi: 10.3390/jfb13020040
- Chen Y, Chen H, Harker A, Liu Y, Huang J. A supervised machine learning tool to predict the bactericidal efficiency of nanostructured surface. J Nanobiotechnol. 2024;22(1):748. doi: 10.1186/s12951-024-02974-8
- Chen H, Liu Y, Balabani S, Hirayama R, Huang J. Machine learning in predicting printable biomaterial formulations for direct ink writing. Research (Wash D C). 2023;6:0197. doi: 10.34133/research.0197
- Filippi M, Mekkattu M, Katzschmann RK. Sustainable biofabrication: From bioprinting to AI-driven predictive methods. Trends Biotechnol. 2025;43(2):290-303. doi: 10.1016/j.tibtech.2024.07.002

