The environmental significance of Citrullus lanatus seed husk in biosorption of Pb2+, Ni2+, and Co2+ from municipal wastewaters

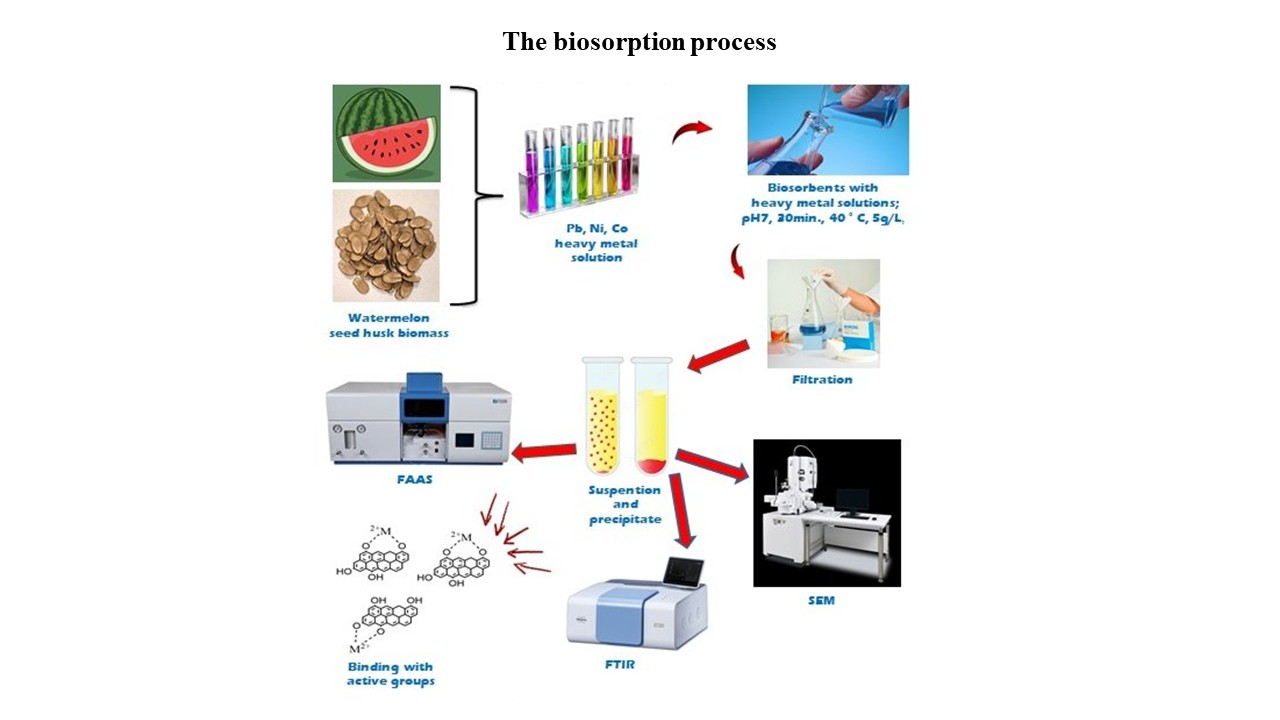
Heavy metal contamination in municipal wastewater poses a serious threat to aquatic ecosystems and human health. This study evaluated the potential of Citrullus lanatus seed husk biomass as a low-cost and sustainable biosorbent for removing lead (Pb2+), nickel (Ni2+), and cobalt (Co2+) ions from wastewater. Batch experiments were conducted to optimize key operational parameters, including pH, temperature, retention time, biosorbent dosage, and particle size. Maximum biosorption efficiencies were achieved at pH near 7, 30–40 °C, and 30 min retention time, with a biosorbent dosage of 5 g/L and a particle size of 0.106 mm. Pb2+ exhibited the highest removal efficiency (up to 95.88%), followed by Ni2+ and Co2+. Fourier transform infrared spectroscopy and scanning electron microscopy analyses confirmed the involvement of hydroxyl and carboxyl functional groups in the adsorption process. Biosorption behavior fits both the Langmuir and the Freundlich isotherm models, indicating favorable and heterogeneous adsorption. Municipal wastewater samples showed unacceptable heavy-metal levels after conventional treatment, whereas post-biosorption levels were reduced to below detectable limits. These findings highlight C. lanatus seed husk as an effective, sustainable, and economically feasible biosorbent for enhancing tertiary wastewater treatment and protecting aquatic ecosystems.
- Beya CB, Kanwugu ON, Ivantsova M. Modern biotechnological methods in wastewater treatment: a review. Chim Techno Acta. 2022;9(2S). doi: 10.15826/chimtech.2022.9.2.S3
- Shartooh SM, Najeeb LM, Sirhan MM. Biological treatment of carcinogenic acrylonitrile using pseudomonas aeruginosa in Basra city. J Biol Sci. 2018;18(8):415-424. doi: 10.3923/jbs.2018.415.424
- Abujazara MS, Abu Amr SS, Alazaiza MY, Albahnasawi A, Ramu MB. Heavy metal removal from industrial wastewater using Pinecone seed powder and Aluminum Chloride- based hybrid natural/chemical coagulation. Water Conserv Manag. 2024;9(1):1-6. doi: 10.26480/wcm.01.2025.49.54
- Bhattacharjee C, Dutta S, Saxena VK. A review on biosorptive removal of dyes and heavy metals from wastewater using watermelon rind as biosorbents. Environ Adv. 2020;2:100007. doi: 10.1016/j.envadv.2020.100007
- Chen RJ, Lee VR. Cobalt Toxicity. In: StatPearls. StatPearls Publishing; 2026. Available from: https://www.ncbi.nlm.nih.gov/books/NBK587403/ [Last accessed on 2025 Oct 15].
- Briffa J, Sinagra E, Blundell R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon. 2020;6(9):e04691. doi: 10.1016/j.heliyon.2020.e04691
- Abdullah SA. Preparation of poly (Hydroxamic acid) from poly (Methylmethacrylate – Co-Methylmethacrylate) and studying of adsorption capacity for Fe III and Cu II. J Univ Anbar Pure Sci. 2011;5(3):64-73. doi: 10.37652/juaps.2011.44282
- Salih SM. A Review on Factors Affecting the Adsorption of Heavy Metal Using Different Biosorbents. Anbar J Eng Sci. 2024;15(1):25-40. doi: 10.37649/aengs.2024.145517.1068
- Li J, Meng X, Song M, Xue M. Enhanced adsorption desulfurization performance of metal-modified Y zeolites. Asian J Water Environ Pollut. 2025;22(6):89-102. doi: 10.36922/AJWEP025250204
- Duwiejuah AB, Mutawakil Z, Oyelude EO. Eco-friendly banana peel biochar for adsorption of toxic metals from landfill treatment pond leachate. Int J Phytoremediation. 2025;27(5):596-605. doi: 10.1080/15226514.2024.2428434
- Khalfaoui A, Benalia A, Selama Z, et al. Removal of Chromium (VI) from Water Using Orange peel as the Biosorbent: Experimental, Modeling, and Kinetic Studies on Adsorption Isotherms and Chemical Structure. Water. 2024;16(5):742. doi: 10.3390/w16050742
- Kwikima MM, Mateso S, Chebude Y. Potentials of agricultural wastes as the ultimate alternative adsorbent for cadmium removal from wastewater. A review. Sci Afr. 2021;13:e00934. doi: 10.1016/j.sciaf.2021.e00934
- Aljeboree AM, Hamid GS, Katham AA, et al. Highly adsorbent surface from watermelon peels: as non- conventional low-cost sorbent; Equilibrium and Recycle study. IOP Conf Ser Earth Environ Sci. 2022;1029:012008. doi: 10.1088/1755-1315/1029/1/012008
- Fertu DI, Bulgariu L, Gavrilescu M. Modeling and Optimization of Heavy Metals Biosorption by Low-Cost Sorbents Using Response Surface Methodology. Processes. 2022;10(3):523. doi: 10.3390/pr10030523
- Alafnan S, Abeeb A, Guenther G, Adjei S, Alrumaih I, Gowida A. Langmuir adsorption isotherm in unconventional resources: Applicability and limitations. J Pet Sci Eng. 2021;207:109172. doi: 10.1016/j.petrol.2021.109172
- Mousavi SM, Hashemi SA, Amani AM, et al. Pb(II) removal from synthetic wastewater using Kombucha Scoby and graphene oxide/Fe3O4. Phys Chem Res. 2018;6(4):759-771. doi: 10.22036/pcr.2018.133392.1490
- Tran HN. Improper estimation of thermodynamic parameters in adsorption studies with distribution coefficient KD (qe/Ce) or Freundlich constant (KF): considerations from the derivation of dimensionless thermodynamic equilibrium constant and suggestions. Adsorpt Sci Technol. 2022;2022:1-23. doi: 10.1155/2022/5553212
- Musah M, Azeh Y, Mathew JT, Tanko MU, Abdulhamid Z, Muhammad AI. Adsorption kinetics and isotherm models: a review. Caliphate J Sci Technol. 2022;4(1):20-26. doi: 10.4314/cajost.v4i1.3
- Rajahmundry GK, Garlapati C, Kumar PS, Alwi RS, Vo DN. Statistical analysis of adsorption isotherm models and its appropriate selection. Chemosphere. 2021;276:130176. doi: 10.1016/j.chemosphere.2021.130176
- Balarak D, Baniasadi M, Lee SM, Shim MJ. Ciprofloxacin adsorption onto Azolla filiculoides activated carbon from aqueous solutions. Desalin Water Treat. 2021;218:444-453. doi: 10.5004/dwt.2021.26986
- Karim MA, Ariyanto E, Juniar H, Astuti PW. Kinetic, isotherm, and thermodynamic study of Cu(II) ion removal from groundwater using acid-activated carbon based on coconut tree waste (Cocos nucifera L.). Ecol Eng Environ Technol. 2025;26(7):1-16. doi: 10.12912/27197050/204508
- Naddaf M. A Study of Some Chemical Components of Watermelon Seeds planted in Syria and their Oil Characteristics and Composition. Tishreen Univ J Res Sci Stud. 2008;30(2):27-39.
- Onyango C, Nyairo W, Tchieta GP, Shikuku VO. Watermelon rind based adsorbents for the removal of water pollutants: a critical review. Front Environ Chem. 2025;6:1568695. doi: 10.3389/fenvc.2025.1568695
- Elaine E, Jess WT, Nyam KL. Watermelon seeds: nutritional profile, bioactivities, and application in food industry. In: Valorization of Fruit Seed Waste from Food Processing Industry. 2024:183-213. doi: 10.1016/B978-0-443-15535-2.00004-9
- Al-Hazeef MSF, Aidi A, Hecini L, et al. Valorizing date palm spikelets into activated carbon-derived composite for methyl orange adsorption: advancing circular bioeconomy in wastewater treatment—a comprehensive study on its equilibrium, kinetics, thermodynamics, and mechanisms. Environ Sci Pollut Res. 2024;31(38):50493- 50512. doi: 10.1007/s11356-024-34581-3
- Boumchita S, Lahrichi A, Benjelloum Y, Lairini S, Nenov V, Zerrouq F. Application of Peanut shell as a low-cost adsorbent for the removal of anionic dye from aqueous solutions. J Mater Environ Sci. 2017;8(7):2353-2364. Available from: https://www.researchgate.net/publication/318305907_ Application_of_peanut_shell_as_a_low-cost_adsorbent_for_the_removal_of_anionic_dye_from_aqueous_solutions [Last accessed on 2025 Oct 1].
- Stadnik J, Giusti ED, Borges AR. Investigation of the zero charge point of orange biomass for further use as a metal biosorbent. In: A Look at Development. Seven Editora; 2023. doi: 10.56238/alookdevelopv1-103
- Kumari B, Dutta S. Integrating starch encapsulated nanoscale zero-valent iron for better Chromium removal performance. J Water Process Eng. 2020;37:101370. doi: 10.1016/j.jwpe.2020.101370
- Rahman AMF, Suhaimi MAA, Othman N. Biosorption of Pb (II) and Zn (II) in synthetic wastewater by Watermelon rind (Citrullus lanatus). Appl Mech Mater. 2014;465-466:906- 910. doi: 10.4028/www.scientific.net/AMM.465-466.906
- Lakshmipathy R, Sarada NC. Metal ion free watermelon (Citrullus lanatus) rind as adsorbent for the removal of lead and copper ions from aqueous solution. Desalin Water Treat. 2016;57(33):15362-15372. doi: 10.1080/19443994.2015.1072064
- Lakshmipathy R, Reddy NA, Sarada NC. Optimization of brilliant green biosorption by native and acid-activated watermelon rind as low-cost adsorbent. Desalin Water Treat. 2015;54(1):235-244. doi: 10.1080/19443994.2013.879082
- Altowayti WAH, Othman N, Al-Gheethi A, et al. Adsorption of Zn2+ from Synthetic Wastewater Using Dried Watermelon Rind (D-WMR): An Overview of Nonlinear and Linear Regression and Error Analysis. Molecules. 2021;26(20):6176. doi: 10.3390/molecules26206176
- Archibong UD, Ikpe EE, Amienghemhen OD. Adsorption of copper and cadmium from wastewater using chemically activated watermelon peels as adsorbent. Chem Sci Int J. 2024;33(1):1-11. doi: 10.9734/CSJI/2024/v33i1879
- Okam EC, Odo GI, Uduma CK, Ijioma GU, Okolo BI. Biosorption of chromium (II) ion from textile effluent using watermelon shell-activated carbon. Path Sci. 2022;8(9):1018- 1029. doi: 10.22178/pos.85-2
- Torres E. Biosorption: A Review of the Latest Advances. Processes. 2020;8(12):1584. doi: 10.3390/pr8121584
- Wang Q, Zhu S, Xi C, Zhang F. A Review: Adsorption and Removal of Heavy Metals Based on Polyamide-amines Composites. Front Chem. 2022;10:814643. doi: 10.3389/fchem.2022.814643
- Nyairo W, Njewa JB, Shikuku VO. Adsorption of heavy metals onto food wastes: a review. Front Environ Chem. 2025;6:1526366. doi: 10.3389/fenvc.2025.1526366
- Sharma A, Devi I. Biosorption of heavy metal ions from aqueous effluents utilising snail shell dust as a biomaterial. Environ Dev Sustain. 2024;26(12):31879-31896. doi: 10.1007/s10668-024-04777-1
- Flores-Trujillo AKI, Mussali-Galante P, de Hoces MC, et al. Biosorption of heavy metals on Opuntia fuliginosa and Agave angustifolia fibers for their elimination from water. Int J Environ Sci Technol. 2021;18(2):441-454. doi: 10.1007/s13762-020-02832-8
- Khazaal ShH, Al-Sheikh F, Al-Ameri M. Using activated carbon to adsorb Co (II) from synthetic solution: Isotherms and optimization studies. AIP Conf Proc. 2022;2670:060017. doi: 10.1063/5.0095860
- Obaid SA. Langmuir, Freundlich and Tamkin adsorption isotherms and kinetics for the removal Aartichoke tournefortii straw from agricultural waste. J Phys Conf Ser. 2020;1664(1):012011. doi: 10.1088/1742-6596/1664/1/012011
- Maharjan J, Jha VK. Activated carbon obtained from banana peels for the removal of As(III) from water. Sci World. 2022;15(15):145-157. doi: 10.3126/sw.v15i15.45665
- Jasper EE, Ajibola VO, Onwuka JC. Nonlinear regression analysis of the sorption of crystal violet and methylene blue from aqueous solutions onto an agro-waste derived activated carbon. Appl Water Sci. 2020;10(6):132. doi: 10.1007/s13201-020-01218-y
- Hicham A, Hussein J, Siba H. Kinetic, isotherm and thermodynamic studies on the ciprofloxacin adsorption from aqueous solution using Aleppo bentonite. Baghdad Sci J. 2022;19(3):680-692. doi: 10.21123/bsj.2022.19.3.0680
- Guidelines for Drinking Water Quality. 4th ed. Geneva, Switzerland: WHO Press; 2011. Available from: https://www.who.int/publications/i/item/9789241549950 [Last accessed on 2025 Aug 25].
- Essa AK, Althwani MN, Dawood TN. Estimate the level of some of heavy metals in treated recurrent sewage water from Rustimayah station to Dyala River. J Fac Basic Educ. 2012;75:221-235. doi: 10.35950/cbej.v18i75.9230

