Laponite-reinforced conductive GelMA–-Ionic liquid nanocomposite hydrogels for high-fidelity extrusion printing and localized neurotrophic delivery

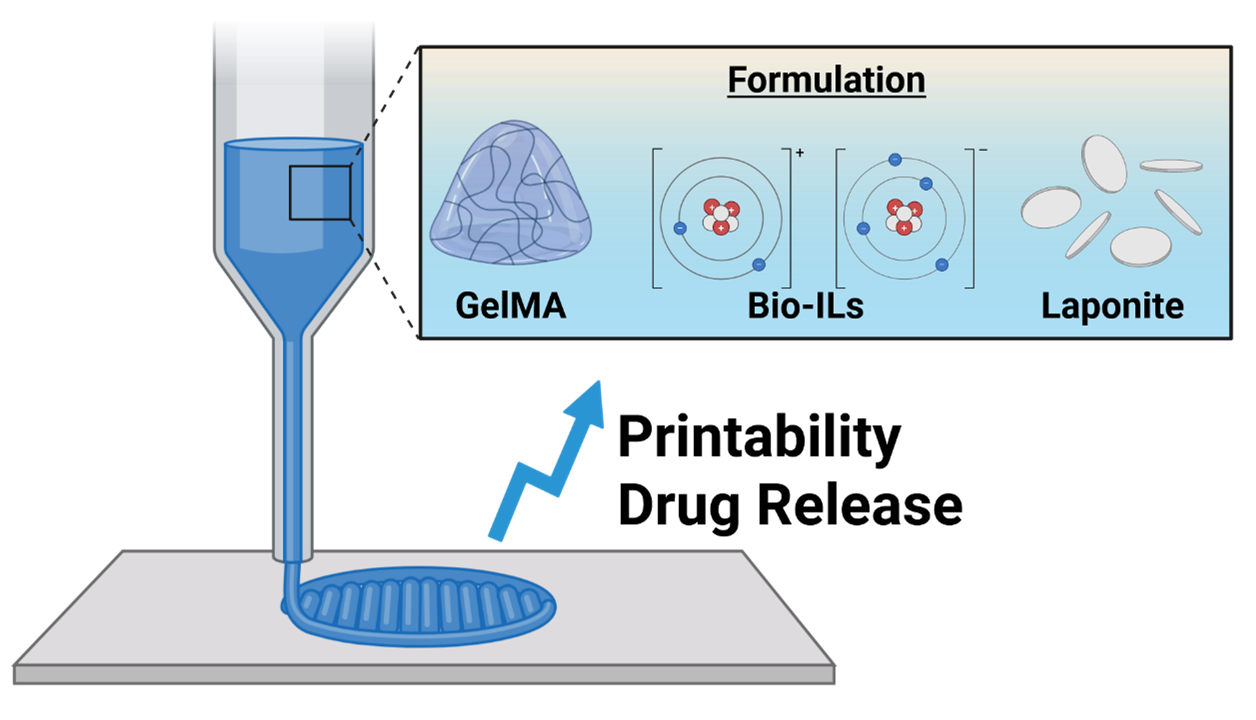
The development of advanced hydrogel systems capable of precise three dimensional (3D) printing and controlled therapeutic delivery is critical for next generation biofabrication strategies. In this study, we present a laponite-reinforced gelatin methacryloyl (GelMA)/ionic liquid hydrogel engineered to simultaneously improve printability and sustained release of bioactive molecules. The incorporation of laponite nanoparticles markedly enhances rheological characteristics, including viscosity, shear-thinning behavior, and structural fidelity, facilitating high-resolution extrusion-based 3D printing. Specifically, the laponite concentration was limited to 1% w/v to preserve the soft mechanical environment (<3 kPa) essential for neural tissue while sufficiently improving rheological properties for processing. Beyond its mechanical and processing advantages, the hydrogel enables the prolonged release of retinoic acid and glial cell line-derived neurotrophic factor, promoting the proliferation and neuronal differentiation of N2A cells. This dual-functional platform demonstrates significant potential for the fabrication of complex, cell-instructive scaffolds, offering a versatile approach for applications where structural precision and localized drug delivery are essential.
- Vijayavenkataraman S, Yan W-C, Lu WF, Wang C-H, Fuh JYH. 3D bioprinting of tissues and organs for regenerative medicine. Adv Drug Deliv Rev. 2018;132:296-332. doi: 10.1016/j.addr.2018.07.004
- Mani MP, Sadia M, Jaganathan SK, et al. A review on 3D printing in tissue engineering applications. J Polym Eng. 2022;42(3):243-265. doi: 10.1515/polyeng-2021-0059
- Saini G, Segaran N, Mayer J, Saini A, Albadawi H, Oklu R. Applications of 3D Bioprinting in Tissue Engineering and Regenerative Medicine. J Clin Med. 2021;10(21):4966. doi: 10.3390/jcm10214966
- Acierno D, Patti A. Fused Deposition Modelling (FDM) of Thermoplastic-Based Filaments: Process and Rheological Properties—An Overview. Materials. 2023;16(24):7664. doi: 10.3390/ma16247664
- Ferretti P, Santi GM, Leon-Cardenas C, Fusari E, Cristofori M, Liverani A. Production readiness assessment of low cost, multi-material, polymeric 3D printed moulds. Heliyon. 2022;8(10):e11136. doi: 10.1016/j.heliyon.2022.e11136
- Sood AK, Ohdar RK, Mahapatra SS. Experimental investigation and empirical modelling of FDM process for compressive strength improvement. J Adv Res. 2012;3(1):81-90. doi: 10.1016/j.jare.2011.05.001
- Li J, Wu C, Chu PK, Gelinsky M. 3D printing of hydrogels: Rational design strategies and emerging biomedical applications. Mater Sci Eng R Rep. 2020;140:100543. doi: 10.1016/j.mser.2020.100543
- Herrada-Manchon H, Fernandez MA, Aguilar E. Essential Guide to Hydrogel Rheology in Extrusion 3D Printing: How to Measure It and Why It Matters? Gels. 2023;9(7):517. doi: 10.3390/gels9070517
- He Y, Yang F, Zhao H, Gao Q, Xia B, Fu J. Research on the printability of hydrogels in 3D bioprinting. Sci Rep. 2016;6(1)doi:10.1038/srep29977
- Leu Alexa R, Iovu H, Ghitman J, et al. 3D-Printed Gelatin Methacryloyl-Based Scaffolds with Potential Application in Tissue Engineering. Polymers. 2021;13(5):727. doi: 10.3390/polym13050727
- Aldana AA, Valente F, Dilley R, Doyle B. Development of 3D bioprinted GelMA-alginate hydrogels with tunable mechanical properties. Bioprinting. 2021;21:e00105. doi: 10.1016/j.bprint.2020.e00105
- Ding H, Illsley NP, Chang RC. 3D Bioprinted GelMA Based Models for the Study of Trophoblast Cell Invasion. Sci Rep. 2019;9(1). doi: 10.1038/s41598-019-55052-7
- Grosskopf AK, Saouaf OA, Lopez Hernandez H, Appel EA. Gelation and yielding behavior of polymer–nanoparticle hydrogels. J Polym Sci. 2021;59(22):2854-2866. doi: 10.1002/pol.20210652
- Xu P, Shang Z, Yao M, Li X. Mechanistic insight into improving strength and stability of hydrogels via nanosilica. J Mol Liq. 2022;357:119094. doi: 10.1016/j.molliq.2022.119094
- Yoon J, Han H, Jang J. Nanomaterials-incorporated hydrogels for 3D bioprinting technology. Nano Converg. 2023/11/15 2023;10(1) doi: 10.1186/s40580-023-00402-5
- Dong L, Bu Z, Xiong Y, et al. Facile extrusion 3D printingof gelatine methacrylate/Laponite nanocomposite hydrogel with high concentration nanoclay for bone tissue regeneration. Int J Biol Macromol. 2021;188:72-81. doi: 10.1016/j.ijbiomac.2021.07.199
- Dawson JI, Kanczler JM, Yang XB, Attard GS, Oreffo ROC. Clay Gels For the Delivery of Regenerative Microenvironments. Adv Mater. 2011/06/10 2011;23(29):3304-3308. doi: 10.1002/adma.201100968
- Dawson JI, Oreffo ROC. Clay: New Opportunities for Tissue Regeneration and Biomaterial Design. Adv Mater. 2013;25(30):4069-4086. doi: 10.1002/adma.201301034
- Cidonio G, Glinka M, Kim Y-H, et al. Nanoclay-based 3D printed scaffolds promote vascular ingrowth ex vivo and generate bone mineral tissue in vitro and in vivo. Biofabrication. 2020/05/11 2020;12(3):035010. doi: 10.1088/1758-5090/ab8753
- Cidonio G, Alcala-Orozco CR, Lim KS, et al. Osteogenic and angiogenic tissue formation in high fidelity nanocomposite Laponite-gelatin bioinks. Biofabrication. 2019;11(3):035027. doi: 10.1088/1758-5090/ab19fd
- Tan Y, Xu S, Wu R, Du J, Sang J, Wang J. A gradient Laponite crosslinked nanocomposite hydrogel with anisotropic stress and thermo-response. Appl Clay Sci. 2017;148:77-82. doi: 10.1016/j.clay.2017.08.004
- Sheikhi A, Afewerki S, Oklu R, Gaharwar AK, Khademhosseini A. Effect of ionic strength on shearthinning nanoclay–polymer composite hydrogels. Biomater Sci. 2018;6(8):2073-2083. doi: 10.1039/c8bm00469b
- Ruzicka B, Zaccarelli E. A fresh look at the Laponite phase diagram. Soft Matter. 2011;7(4):1268. doi: 10.1039/c0sm00590h
- Ng WL, Yeow CHE, Huang X, Lee S, Yeong WY. Physically cross-linked gelatin bio-inks with enhanced printability, degradation and mechanical robustness for multi-modal bioprinting. Interdiscip Med. 2025;3(4):e20250058. doi: 10.1002/INMD.20250058
- Gunduz O, Ustundag CB, Sengor M, editors. Biomaterials for Neural Tissue Engineering. Elsevier; 2023.
- Elkwood AI, Holland NR, Arbes SM, et al. Nerve allograft transplantation for functional restoration of the upper extremity: case series. J Spinal Cord Med. 2011;34(2):241-247. doi: 10.1179/107902611x12972448729521
- Singh VK, Haq A, Tiwari M, Saxena AK. Approach to management of nerve gaps in peripheral nerve injuries. Injury. 2022;53(4):1308-1318. doi: 10.1016/j.injury.2022.01.031
- Supra R, K Agrawal D. Peripheral Nerve Regeneration: Opportunities and Challenges. J Spine Res Surg. 2023;05(01). doi: 10.26502/fjsrs0052
- Zhou J, Vijayavenkataraman S. 3D-printable conductive materials for tissue engineering and biomedical applications.Bioprinting. 2021;24:e00166. doi: 10.1016/j.bprint.2021.e00166
- Carvalho CR, Chang W, Silva‐Correia J, Reis RL, Oliveira JM, Kohn J. Engineering Silk Fibroin‐Based Nerve Conduit with Neurotrophic Factors for Proximal Protection after Peripheral Nerve Injury. Adv Healthc Mater. 2020;10(2). doi: 10.1002/adhm.202000753
- Nectow AR, Marra KG, Kaplan DL. Biomaterials for the Development of Peripheral Nerve Guidance Conduits. Tissue Eng Part B Rev. 2012;18(1):40-50. doi: 10.1089/ten.teb.2011.0240
- Zhao Y, Liang Y, Ding S, Zhang K, Mao HQ, Yang Y. Application of conductive PPy/SF composite scaffold and electrical stimulation for neural tissue engineering. Biomaterials. 2020;255:120164. doi: 10.1016/j.biomaterials.2020.120164
- Fukaya Y, Iizuka Y, Sekikawa K, Ohno H. Bio ionic liquids: room temperature ionic liquids composed wholly of biomaterials. Green Chem. 2007;9(11):1155. doi: 10.1039/b706571j
- Kuchenbuch A, Giernoth R. Ionic Liquids Beyond Simple Solvents: Glimpses at the State of the Art in Organic Chemistry. ChemistryOpen. 2015;4(6):677-681. doi: 10.1002/open.201500113
- Lei Z, Chen B, Koo YM, MacFarlane DR. Introduction: Ionic Liquids. Chem Rev. 2017;117(10):6633-6635. doi: 10.1021/acs.chemrev.7b00246
- Kuchenbuch A, Giernoth R. Ionic Liquids Beyond Simple Solvents: Glimpses at the State of the Art in Organic Chemistry. ChemistryOpen. 2015;4(6):677-681. doi: 10.1002/open.201500113
- Gomes JM, Silva SS, Reis RL. Biocompatible ionic liquids: fundamental behaviours and applications. Chem Soc Rev. 2019;48(15):4317-4335. doi: 10.1039/c9cs00016j
- Noshadi I, Walker BW, Portillo-Lara R, et al. Engineering Biodegradable and Biocompatible Bio-ionic Liquid Conjugated Hydrogels with Tunable Conductivity and Mechanical Properties. Sci Rep. 2017;7(1):4345. doi: 10.1038/s41598-017-04280-w
- Krishnadoss V, Kanjilal B, Masoumi A, et al. Programmable bio-ionic liquid functionalized hydrogels for in situ 3D bioprinting of electronics at the tissue interface. Mater Today Adv. 2023;17:100352. doi: 10.1016/j.mtadv.2023.100352
- Zhou J, Elkhoury K, Saghar Soman S, Vijayavenkataraman S. Biofabrication of tri-layered nerve guide conduits for peripheral nerve regeneration: Synergizing melt electrowriting of polymeric fibers and extrusion-based 3D bioprinting. Int J Bioprinting. 2025. doi: 10.36922/ijb025040032
- Menon A, Elkhoury K, Zahraa A, et al. Digital light processing 3D printing of dual cross-linked meniscal scaffolds with enhanced physical and biological properties. Adv Compos Hybrid Mater. 2024;8(1). doi: 10.1007/s42114-024-01196-8
- Saha K, Keung AJ, Irwin EF, et al. Substrate Modulus Directs Neural Stem Cell Behavior. Biophys J. 2008;95(9):4426-4438. doi: 10.1529/biophysj.108.132217
- Seidlits SK, Khaing ZZ, Petersen RR, et al. The effects of hyaluronic acid hydrogels with tunable mechanical properties on neural progenitor cell differentiation. Biomaterials. 2010;31(14):3930-3940. doi: 10.1016/j.biomaterials.2010.01.125
- Liu J, Zheng H, Poh P, Machens H-G, Schilling A. Hydrogels for Engineering of Perfusable Vascular Networks. Int J Mol Sci. 2015/07/14 2015;16(7):15997-16016. doi: 10.3390/ijms160715997
- Davern JW, Hipwood L, Bray LJ, Meinert C, Klein TJ. Addition of Laponite to gelatin methacryloyl bioinks improves the rheological properties and printability to create mechanically tailorable cell culture matrices. APL Bioeng. 2024/01/08 2024;8(1). doi: 10.1063/5.0166206
- Ma Y, Wang X, Su T, Lu F, Chang Q, Gao J. Recent Advances in Macroporous Hydrogels for Cell Behavior and Tissue Engineering. Gels. 2022/09/21 2022;8(10):606. doi: 10.3390/gels8100606
- Annabi N, Nichol JW, Zhong X, et al. Controlling the Porosity and Microarchitecture of Hydrogels for Tissue Engineering. Tissue Eng Part B Rev. 2010;16(4):371-383. doi: 10.1089/ten.teb.2009.0639
- Zeltinger J, Sherwood JK, Graham DA, Mueller R, Griffith LG. Effect of Pore Size and Void Fraction on Cellular Adhesion, Proliferation, and Matrix Deposition. Tissue Eng. 2001;7(5):557-572. doi: 10.1089/107632701753213183
- Žigon-Branc S, Markovic M, Van Hoorick J, et al. Impact of Hydrogel Stiffness on Differentiation of Human Adipose-Derived Stem Cell Microspheroids. Tissue Eng Part A. 2019;25(19-20):1369-1380. doi: 10.1089/ten.tea.2018.0237
- Holloway JL, Ma H, Rai R, Burdick JA. Modulating hydrogel cross-link density and degradation to control bone morphogenetic protein delivery and in vivo bone formation. J. Control Release. 2014;191:63-70. doi: 10.1016/j.jconrel.2014.05.053
- Rambhia KJ, Ma PX. Controlled drug release for tissue engineering. J Control Release. 2015;219:119-128. doi: 10.1016/j.jconrel.2015.08.049
- Li YM, Ji Y, Meng YX, et al. Neural Tissue-Like, not Supraphysiological, Electrical Conductivity Stimulates Neuronal Lineage Specification through Calcium Signaling and Epigenetic Modification. Adv Sci. 2024;11(35):e2400586. doi: 10.1002/advs.202400586
- Chakraborty J, Majumder N, Sharma A, Prasad S, Ghosh S. 3D bioprinted silk-reinforced Alginate-Gellan Gum constructs for cartilage regeneration. Bioprinting. 2022;28:e00232. doi: 10.1016/j.bprint.2022.e00232
- Munoz-Perez E, Perez-Valle A, Igartua M, Santos-Vizcaino E, Hernandez RM. High resolution and fidelity 3D printing of Laponite and alginate ink hydrogels for tunable biomedical applications. Biomater Adv. 2023;149:213414. doi: 10.1016/j.bioadv.2023.213414
- Ghadiri M, Chrzanowski W, Rohanizadeh R. Antibiotic eluting clay mineral (LaponiteR) for wound healing application: an in vitro study. J Mater Sci Mater Med. 2014;25(11):2513-2526. doi: 10.1007/s10856-014-5272-7
- Kiaee G, Mostafalu P, Samandari M, Sonkusale S. A Ph Mediated Electronic Wound Dressing for Controlled Drug Delivery. Adv Healthc Mater. 2018;7(18). doi: 10.1002/adhm.201800396
- Wang C, Gong Z, Huang X, et al. An injectable heparin-Laponite hydrogel bridge FGF4 for spinal cord injury by stabilizing microtubule and improving mitochondrial function. Theranostics. 2019;9(23):7016-7032. doi: 10.7150/thno.37601
- Wang G, Maciel D, Wu Y, et al. Amphiphilic Polymer- Mediated Formation of Laponite-Based Nanohybrids with Robust Stability and pH Sensitivity for Anticancer Drug Delivery. ACS Appl Mater Interfaces. 2014;6(19):16687-16695. doi: 10.1021/am5032874
- Zhang M, Bai Y, Xu C, et al. Novel optimized drug delivery systems for enhancing spinal cord injury repair in rats. Drug Deliv. 2021;28(1):2548-2561. doi: 10.1080/10717544.2021.2009937
- Zhou B, Wu B, Wang J, et al. Drug-mediation formation of nanohybrids for sequential therapeutic delivery in cancer cells. Colloids Surf. B Biointerfaces. 2018;163:284-290. doi: 10.1016/j.colsurfb.2017.12.046
- Cai Y, Chen Y, Zhang G, et al. The GDNF-gel/HA-Mg conduit promotes the repair of peripheral nerve defects by regulating PPAR-γ/RhoA/ROCK signaling pathway. iScience. 2024;27(2):108969. doi: 10.1016/j.isci.2024.108969
- Qiu S, Rao Z, He F, et al. Decellularized nerve matrix hydrogel and glial‐derived neurotrophic factor modifications assisted nerve repair with decellularized nerve matrix scaffolds. J Tissue Eng Regen Med. 2020;14(7):931-943. doi: 10.1002/term.3050
- Hosseinzadeh B, Ahmadi M. Degradable hydrogels: Design mechanisms and versatile applications. Mater Today Sustain. 2023;23:100468. doi: 10.1016/j.mtsust.2023.100468
- Patel G, Dalwadi C. Recent Patents on Stimuli Responsive Hydrogel Drug Delivery System. Recent Pat Drug Deliv Formul. 2013;7(3):206-215. doi: 10.2174/1872211307666131118141600
- Xing MMQ, Zhuang H, Bu S, Hua L, Darabi MA, Cao X. Gelatin-methacrylamide gel loaded with microspheres to deliver GDNF in bilayer collagen conduit promoting sciatic nerve growth. Int J Nanomed. 2016:1383. doi: 10.2147/ijn.s96324
- Fadia NB, Bliley JM, DiBernardo GA, et al. Long-gap peripheral nerve repair through sustained release of a neurotrophic factor in nonhuman primates. Sci Transl Med. 2020;12(527)eaav7753. doi: 10.1126/scitranslmed.aav7753
- Ansary RH, Awang MB, Rahman MM. Biodegradable Poly(D,L-lactic-co-glycolic acid)-Based Micro/Nanoparticles for Sustained Release of Protein Drugs – A Review. Trop J Pharm Res. 2014;13(7):1179. doi: 10.4314/tjpr.v13i7.24
- Blanco D, Alonso MaJ. Protein encapsulation and release from poly(lactide-co-glycolide) microspheres: effect of the protein and polymer properties and of the co-encapsulation of surfactants. Eur J Pharm Biopharm. 1998;45(3):285-294. doi: 10.1016/s0939-6411(98)00011-3
- Estey T, Kang J, Schwendeman SP, Carpenter JF. BSA Degradation Under Acidic Conditions: A Model For Protein Instability During Release From PLGA Delivery Systems. J Pharm Sci. 2006;95(7):1626-1639. doi: 10.1002/jps.20625
- Gavali KV, Kengar MD, Chavan KV, Anekar VP, Khan NI. A Review on Microsphere and it’s Application. Asian J Pharm Res. 2019;9(2):123. doi: 10.5958/2231-5691.2019.00020.0
- Kang J, Schwendeman SP. Comparison of the effects of Mg(OH)2 and sucrose on the stability of bovine serum albumin encapsulated in injectable poly(d,l-lactide-coglycolide) implants. Biomaterials. 2002;23(1):239-245. doi: 10.1016/s0142-9612(01)00101-6
- Lin L-FH, Doherty DH, Lile JD, Bektesh S, Collins F. GDNF: a Glial Cell Line-Derived Neurotrophic Factor for Midbrain Dopaminergic Neurons. Science. 1993;260(5111):1130- 1132. doi: 10.1126/science.8493557
- Zhang L, Ma Z, Smith GM, et al. GDNF‐enhanced axonal regeneration and myelination following spinal cord injury is mediated by primary effects on neurons. Glia. 2009;57(11):1178-1191. doi: 10.1002/glia.20840
- Xu P, Rosen KM, Hedstrom K, et al. Nerve injury induces glial cell line‐derived neurotrophic factor (gdnf) expression in schwann cells through purinergic signaling and the pkcpkd pathway. Glia. 2013;61(7):1029-1040. doi: 10.1002/glia.22491
- Hoke A, Gordon T, Zochodne DW, Sulaiman OAR. A decline in glial cell-line-derived neurotrophic factor expression is associated with impaired regeneration after long-term Schwann cell denervation. Article. Exp Neurol. 2002;173(1):77-85. doi: 10.1006/exnr.2001.7826
- Airaksinen MS, Saarma M. The GDNF family: Signalling, biological functions and therapeutic value. Nat Rev Neurosci. 2002;3(5):383-394. doi: 10.1038/nrn812
- Yue P, Gao L, Wang X, Ding X, Teng J. Intranasal Administration of GDNF Protects Against Neural Apoptosis in a Rat Model of Parkinson’s Disease Through PI3K/Akt/GSK3β Pathway. Neurochem Res. 2017;42(5):1366-1374. doi: 10.1007/s11064-017-2184-1
- Kumar M, Katyal A. Data on retinoic acid and reduced serum concentration induced differentiation of Neuro-2a neuroblastoma cells. Data Brief. 2018;21:2435-2440. doi: 10.1016/j.dib.2018.11.097
- Namsi A, Nury T, Hamdouni H, et al. Induction of Neuronal Differentiation of Murine N2a Cells by Two Polyphenols Present in the Mediterranean Diet Mimicking Neurotrophins Activities: Resveratrol and Apigenin. Diseases. 2018;6(3):67. doi: 10.3390/diseases6030067
- Shimizu S, Kondo M, Miyamoto Y, Hayashi M. Foxa(HNF3) Upregulates Vitronectin Expression during Retinoic Acid induced Differentiation in Mouse Neuroblastoma Neuro2a Cells. Cell Struct Funct. 2002;27(4):181-188. doi: 10.1247/csf.27.181
- Du Y-J, Dong S-Z, You Q, Gong Q, Han Y-Q, Pi R. Role of miR-124 in the regulation of retinoic acid-induced Neuro-2A cell differentiation. Neural Regen Res. 2020;15(6):1133. doi: 10.4103/1673-5374.270417
- Runeberg-Roos P, Penn RD. Improving therapeutic potential of GDNF family ligands. Cell Tissue Res. 2020;382(1):173-183. doi: 10.1007/s00441-020-03256-z
- Esteves M, Cristovao AC, Saraiva T, et al. Retinoic acid loaded polymeric nanoparticles induce neuroprotection in a mouse model for Parkinson’s disease. Front Aging Neurosci. 2015;7.doi: 10.3389/fnagi.2015.00020
- Ye T, Pollack GH. Do aqueous solutions contain net charge? PLOS ONE. 2022/10/27 2022;17(10):e0275953. doi: 10.1371/journal.pone.0275953
- Rodrigo MJ, Cardiel MJ, Fraile JM, Mayoral JA, Pablo LE, Garcia-Martin E. Laponite for biomedical applications: An ophthalmological perspective. Materials Today Bio. 2024;24:100935. doi: 10.1016/j.mtbio.2023.100935
- Hu H, Dong L, Bu Z, et al. miR‐23a‐3p‐abundant small extracellular vesicles released from Gelma/nanoclay hydrogel for cartilage regeneration. J Extracell Vesicles. 2020;9(1). doi: 10.1080/20013078.2020.1778883
- Man K, Barroso IA, Brunet MY, et al. Controlled Release of Epigenetically-Enhanced Extracellular Vesicles from a GelMA/Nanoclay Composite Hydrogel to Promote Bone Repair. Int J Mol Sci. 2022;23(2):832. doi: 10.3390/ijms23020832
- Korzynska A, Zychowicz M. A Method of Estimation of the Cell Doubling Time on Basis of the Cell Culture Monitoring Data. Biocybern Biomed Engineering. 2008;28:75-82.
- Martin D, Ruano D, Yufera A, Daza P. Electrical pulse stimulation parameters modulate N2a neuronal differentiation. Cell Death Discov. 2024;10(1). doi: 10.1038/s41420-024-01820-y
- Rago AP, Napolitano AP, Dean DM, Chai PR, Morgan JR. Miniaturization of an Anoikis assay using non-adhesive micromolded hydrogels. Cytotechnology. 2007;56(2):81-90. doi: 10.1007/s10616-007-9116-x
- Yue K, Trujillo-de Santiago G, Alvarez MM, Tamayol A, Annabi N, Khademhosseini A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254-271. doi: 10.1016/j.biomaterials.2015.08.045
- An J, Chen B, Tian D, Guo Y, Yan Y, Yang H. Regulation of Neurogenesis and Neuronal Differentiation by Natural Compounds. Curr Stem Cell Res Ther. 2022;17(8):756-771. doi: 10.2174/1574888X16666210907141447
- Ruijtenberg S, van den Heuvel S. Coordinating cell proliferation and differentiation: Antagonism between cell cycle regulators and cell type-specific gene expression. Cell Cycle. 2016;15(2):196-212. doi: 10.1080/15384101.2015.1120925
- Nasser TIN, Spencer GE. Neurite Outgrowth. In: Reference Module in Biomedical Sciences. Amsterdam, The Netherlands: Elsevier; 2017.
- Meldolesi J. Neurite outgrowth: This process, first discovered by Santiago Ramon y Cajal, is sustained by the exocytosis of two distinct types of vesicles. Brain Res Rev. 2011;66(1-2):246-255. doi: 10.1016/j.brainresrev.2010.06.004
- Paganoni S, Ferreira A. Neurite extension in central neurons: a novel role for the receptor tyrosine kinases Ror1 and Ror2. J Cell Sci. 2005;118(2):433-446. doi: 10.1242/jcs.01622
- Valtorta F, Leoni C. Molecular mechanisms of neurite extension. Philos Trans R. Soc Lond Ser B Biol. Sci. 1999;354(1381):387-394. doi: 10.1098/rstb.1999.0391

